Gamma-enolase: a well-known tumour marker, with a less-known role in cancer
- PMID: 26401126
- PMCID: PMC4577217
- DOI: 10.1515/raon-2015-0035
Gamma-enolase: a well-known tumour marker, with a less-known role in cancer
Abstract
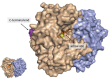
Background: Gamma-enolase, known also as neuron-specific enolase (NSE), is an enzyme of the glycolytic pathway, which is expressed predominantly in neurons and cells of the neuroendocrine system. As a tumour marker it is used in diagnosis and prognosis of cancer; however, the mechanisms enrolling it in malignant progression remain elusive. As a cytoplasmic enzyme gamma-enolase is involved in increased aerobic glycolysis, the main source of energy in cancer cells, supporting cell proliferation. However, different cellular localisation at pathophysiological conditions, proposes other cellular engagements.
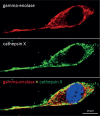
Conclusions: The C-terminal part of the molecule, which is not related to glycolytic pathway, was shown to promote survival of neuronal cells by regulating neuronal growth factor receptor dependent signalling pathways, resulting also in extensive actin cytoskeleton remodelling. This additional function could be important also in cancer cells either to protect cells from stressful conditions and therapeutic agents or to promote tumour cell migration and invasion. Gamma-enolase might therefore have a multifunctional role in cancer progression: it supports increased tumour cell metabolic demands, protects tumour cells from stressful conditions and promotes their invasion and migration.
Keywords: cancer; cell survival; gamma-enolase; glycolysis; tumour marker.
Figures
References
-
- Kim JW, Dang CV. Multifaceted roles of glycolytic enzymes. Trends Biochem Sci. 2005;30:142–50. - PubMed
-
- Masoudi-Nejad A, Asgari Y. Metabolic cancer biology: Structural-based analysis of cancer as a metabolic disease, new sights and opportunities for disease treatment. Semin Cancer Biol. 2015;30:21–9. - PubMed
-
- Dang CV, Semenza GL. Oncogenic alterations of metabolism. Trends Biochem Sci. 1999;24:68–72. - PubMed
-
- McAlister L, Holland MJ. Targeted deletion of a yeast enolase structural gene. Identification and isolation of yeast enolase isozymes. J Biol Chem. 1982;257:7181–8. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

