A Novel Hsp90 Inhibitor Activates Compensatory Heat Shock Protein Responses and Autophagy and Alleviates Mutant A53T α-Synuclein Toxicity
- PMID: 26405178
- PMCID: PMC4658594
- DOI: 10.1124/mol.115.101451
A Novel Hsp90 Inhibitor Activates Compensatory Heat Shock Protein Responses and Autophagy and Alleviates Mutant A53T α-Synuclein Toxicity
Abstract
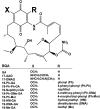
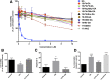
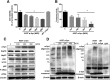
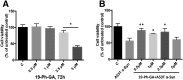
A potential cause of neurodegenerative diseases, including Parkinson's disease (PD), is protein misfolding and aggregation that in turn leads to neurotoxicity. Targeting Hsp90 is an attractive strategy to halt neurodegenerative diseases, and benzoquinone ansamycin (BQA) Hsp90 inhibitors such as geldanamycin (GA) and 17-(allylamino)-17-demethoxygeldanamycin have been shown to be beneficial in mutant A53T α-synuclein PD models. However, current BQA inhibitors result in off-target toxicities via redox cycling and/or arylation of nucleophiles at the C19 position. We developed novel 19-substituted BQA (19BQA) as a means to prevent arylation. In this study, our data demonstrated that 19-phenyl-GA, a lead 19BQA in the GA series, was redox stable and exhibited little toxicity relative to its parent quinone GA in human dopaminergic SH-SY5Y cells as examined by oxygen consumption, trypan blue, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT), and apoptosis assays. Meanwhile, 19-phenyl-GA retained the ability to induce autophagy and potentially protective heat shock proteins (HSPs) such as Hsp70 and Hsp27. We found that transduction of A53T, but not wild type (WT) α-synuclein, induced toxicity in SH-SY5Y cells. 19-Phenyl-GA decreased oligomer formation and toxicity of A53T α-synuclein in transduced cells. Mechanistic studies indicated that mammalian target of rapamycin (mTOR)/p70 ribosomal S6 kinase signaling was activated by A53T but not WT α-synuclein, and 19-phenyl-GA decreased mTOR activation that may be associated with A53T α-synuclein toxicity. In summary, our results indicate that 19BQAs such as 19-phenyl-GA may provide a means to modulate protein-handling systems including HSPs and autophagy, thereby reducing the aggregation and toxicity of proteins such as mutant A53T α-synuclein.
Copyright © 2015 by The American Society for Pharmacology and Experimental Therapeutics.
Figures



Similar articles
-
Caffeic acid reduces A53T α-synuclein by activating JNK/Bcl-2-mediated autophagy in vitro and improves behaviour and protects dopaminergic neurons in a mouse model of Parkinson's disease.Pharmacol Res. 2019 Dec;150:104538. doi: 10.1016/j.phrs.2019.104538. Epub 2019 Nov 9. Pharmacol Res. 2019. PMID: 31707034
-
The mitochondrial chaperone protein TRAP1 mitigates α-Synuclein toxicity.PLoS Genet. 2012 Feb;8(2):e1002488. doi: 10.1371/journal.pgen.1002488. Epub 2012 Feb 2. PLoS Genet. 2012. PMID: 22319455 Free PMC article.
-
Curcumin ameliorates the neurodegenerative pathology in A53T α-synuclein cell model of Parkinson's disease through the downregulation of mTOR/p70S6K signaling and the recovery of macroautophagy.J Neuroimmune Pharmacol. 2013 Mar;8(1):356-69. doi: 10.1007/s11481-012-9431-7. Epub 2013 Jan 17. J Neuroimmune Pharmacol. 2013. PMID: 23325107
-
19-substituted benzoquinone ansamycin heat shock protein-90 inhibitors: biological activity and decreased off-target toxicity.Mol Pharmacol. 2014 Jun;85(6):849-57. doi: 10.1124/mol.113.090654. Epub 2014 Mar 28. Mol Pharmacol. 2014. PMID: 24682466 Free PMC article.
-
Translational Shift of HSP90 as a Novel Therapeutic Target from Cancer to Neurodegenerative Disorders: An Emerging Trend in the Cure of Alzheimer's and Parkinson's Diseases.Curr Drug Metab. 2017;18(9):868-876. doi: 10.2174/1389200218666170728115606. Curr Drug Metab. 2017. PMID: 28758577 Review.
Cited by
-
Geldanamycin, a Naturally Occurring Inhibitor of Hsp90 and a Lead Compound for Medicinal Chemistry.J Med Chem. 2024 Oct 24;67(20):17946-17963. doi: 10.1021/acs.jmedchem.4c01048. Epub 2024 Oct 3. J Med Chem. 2024. PMID: 39361055 Free PMC article. Review.
-
The Molecular Chaperone Artemin Efficiently Blocks Fibrillization of TAU Protein In Vitro.Cell J. 2018 Jan;19(4):569-577. doi: 10.22074/cellj.2018.4510. Epub 2017 Nov 4. Cell J. 2018. PMID: 29105391 Free PMC article.
-
Mammalian/mechanistic target of rapamycin (mTOR) complexes in neurodegeneration.Mol Neurodegener. 2021 Jul 2;16(1):44. doi: 10.1186/s13024-021-00428-5. Mol Neurodegener. 2021. PMID: 34215308 Free PMC article. Review.
-
Neurodegeneration, Mitochondria, and Antibiotics.Metabolites. 2023 Mar 12;13(3):416. doi: 10.3390/metabo13030416. Metabolites. 2023. PMID: 36984858 Free PMC article. Review.
-
Identification of an HSP90 modulated multi-step process for ERBB2 degradation in breast cancer cells.Oncotarget. 2016 Dec 20;7(51):85411-85429. doi: 10.18632/oncotarget.13392. Oncotarget. 2016. PMID: 27863425 Free PMC article.
References
-
- Auluck PK, Chan HYE, Trojanowski JQ, Lee VM-Y, Bonini NM. (2002) Chaperone suppression of alpha-synuclein toxicity in a Drosophila model for Parkinson’s disease. Science 295:865–868. - PubMed
-
- Bové J, Martínez-Vicente M, Vila M. (2011) Fighting neurodegeneration with rapamycin: mechanistic insights. Nat Rev Neurosci 12:437–452. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

