Inhibition of Gli1 mobilizes endogenous neural stem cells for remyelination
- PMID: 26416758
- PMCID: PMC4970518
- DOI: 10.1038/nature14957
Inhibition of Gli1 mobilizes endogenous neural stem cells for remyelination
Abstract
Enhancing repair of myelin is an important but still elusive therapeutic goal in many neurological disorders. In multiple sclerosis, an inflammatory demyelinating disease, endogenous remyelination does occur but is frequently insufficient to restore function. Both parenchymal oligodendrocyte progenitor cells and endogenous adult neural stem cells resident within the subventricular zone are known sources of remyelinating cells. Here we characterize the contribution to remyelination of a subset of adult neural stem cells, identified by their expression of Gli1, a transcriptional effector of the sonic hedgehog pathway. We show that these cells are recruited from the subventricular zone to populate demyelinated lesions in the forebrain but never enter healthy, white matter tracts. Unexpectedly, recruitment of this pool of neural stem cells, and their differentiation into oligodendrocytes, is significantly enhanced by genetic or pharmacological inhibition of Gli1. Importantly, complete inhibition of canonical hedgehog signalling was ineffective, indicating that the role of Gli1 both in augmenting hedgehog signalling and in retarding myelination is specialized. Indeed, inhibition of Gli1 improves the functional outcome in a relapsing/remitting model of experimental autoimmune encephalomyelitis and is neuroprotective. Thus, endogenous neural stem cells can be mobilized for the repair of demyelinated lesions by inhibiting Gli1, identifying a new therapeutic avenue for the treatment of demyelinating disorders.
Figures
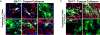





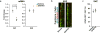







Comment in
-
Repair: Mobilizing stem cells.Nat Rev Neurosci. 2015 Nov;16(11):646. doi: 10.1038/nrn4052. Nat Rev Neurosci. 2015. PMID: 26481096 No abstract available.
References
-
- Xing YL, et al. Adult neural precursor cells from the subventricular zone contribute significantly to oligodendrocyte regeneration and remyelination. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2014;34:14128–14146. doi: 10.1523/JNEUROSCI.3491-13.2014. - DOI - PMC - PubMed
-
- Scolding N, et al. Oligodendrocyte progenitors are present in the normal adult human CNS and in the lesions of multiple sclerosis. Brain: a journal of neurology. 1998;121(Pt 12):2221–2228. - PubMed
-
- Gensert JM, Goldman JE. Endogenous progenitors remyelinate demyelinated axons in the adult CNS. Neuron. 1997;19:197–203. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

