The Vacuolar ATPase a2-subunit regulates Notch signaling in triple-negative breast cancer cells
- PMID: 26418877
- PMCID: PMC4741446
- DOI: 10.18632/oncotarget.5275
The Vacuolar ATPase a2-subunit regulates Notch signaling in triple-negative breast cancer cells
Abstract
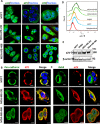
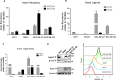
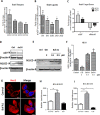
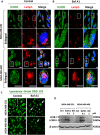
Triple Negative Breast Cancer (TNBC) is a subtype of breast cancer with poor prognosis for which no targeted therapies are currently available. Notch signaling has been implicated in breast cancer but the factors that control Notch in TNBC are unknown. Because the Vacuolar ATPase has been shown to be important in breast cancer invasiveness, we investigated the role of a2-subunit isoform of Vacuolar ATPase (a2V) in regulating Notch signaling in TNBC. Confocal microscopy revealed that among all the 'a' subunit isoforms, a2V was uniquely expressed on the plasma membrane of breast cancer cells. Both a2V and NOTCH1 were elevated in TNBC tumors tissues and cell lines. a2V knockdown by siRNA as well as V-ATPase inhibition by Bafilomycin A1 (Baf A1) in TNBC cell lines enhanced Notch signaling by increasing the expression of Notch1 intracellular Domain (N1ICD). V-ATPase inhibition blocked NICD degradation by disrupting autophagy and lysosomal acidification as demonstrated by accumulation of LC3B and diminished expression of LAMP1 respectively. Importantly, treatment with Baf A1 or anti-a2V, a novel-neutralizing antibody against a2V hindered cell migration of TNBC cells. Our findings indicate that a2V regulates Notch signaling through its role in endolysosomal acidification and emerges as a potential target for TNBC.
Keywords: Notch signaling; a2V-ATPase; autophagy; bafilomycin; triple negative breast cancer.
Conflict of interest statement
The authors declare no conflict of interest
Figures



References
-
- Bandyopadhyay S, Ali-Fehmi R. Breast carcinoma: molecular profiling and updates. Clinics in laboratory medicine. 2013;33:891–909. - PubMed
-
- O'Brien KM, Cole SR, Tse CK, Perou CM, Carey LA, Foulkes WD, Dressler LG, Geradts J, Millikan RC. Intrinsic breast tumor subtypes, race, and long-term survival in the Carolina Breast Cancer Study. Clinical cancer research: an official journal of the American Association for Cancer Research. 2010;16:6100–6110. - PMC - PubMed
-
- O'Toole SA, Beith JM, Millar EK, West R, McLean A, Cazet A, Swarbrick A, Oakes SR. Therapeutic targets in triple negative breast cancer. Journal of clinical pathology. 2013;66:530–542. - PubMed
-
- Mahamodhossen YA, Liu W, Rong-Rong Z. Triple-negative breast cancer: new perspectives for novel therapies. Med Oncol. 2013;30:653. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

