Ty3 Retrotransposon Hijacks Mating Yeast RNA Processing Bodies to Infect New Genomes
- PMID: 26421679
- PMCID: PMC4589538
- DOI: 10.1371/journal.pgen.1005528
Ty3 Retrotransposon Hijacks Mating Yeast RNA Processing Bodies to Infect New Genomes
Abstract
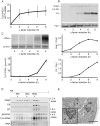
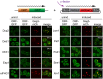
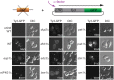
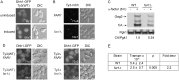
Retrotransposition of the budding yeast long terminal repeat retrotransposon Ty3 is activated during mating. In this study, proteins that associate with Ty3 Gag3 capsid protein during virus-like particle (VLP) assembly were identified by mass spectrometry and screened for roles in mating-stimulated retrotransposition. Components of RNA processing bodies including DEAD box helicases Dhh1/DDX6 and Ded1/DDX3, Sm-like protein Lsm1, decapping protein Dcp2, and 5' to 3' exonuclease Xrn1 were among the proteins identified. These proteins associated with Ty3 proteins and RNA, and were required for formation of Ty3 VLP retrosome assembly factories and for retrotransposition. Specifically, Dhh1/DDX6 was required for normal levels of Ty3 genomic RNA, and Lsm1 and Xrn1 were required for association of Ty3 protein and RNA into retrosomes. This role for components of RNA processing bodies in promoting VLP assembly and retrotransposition during mating in a yeast that lacks RNA interference, contrasts with roles proposed for orthologous components in animal germ cell ribonucleoprotein granules in turnover and epigenetic suppression of retrotransposon RNAs.
Conflict of interest statement
Because the GM33281 grant which supported this work was not renewed publication was paid for by GBB endowed chair to SS. KN is employed by SAIC-Frederick Inc. All other authors have declared that they have no competing interests.
Figures
Similar articles
-
P-body components are required for Ty1 retrotransposition during assembly of retrotransposition-competent virus-like particles.Mol Cell Biol. 2010 Jan;30(2):382-98. doi: 10.1128/MCB.00251-09. Epub 2009 Nov 9. Mol Cell Biol. 2010. PMID: 19901074 Free PMC article.
-
The TY3 Gag3 spacer controls intracellular condensation and uncoating.J Virol. 2011 Apr;85(7):3055-66. doi: 10.1128/JVI.01055-10. Epub 2011 Jan 26. J Virol. 2011. PMID: 21270167 Free PMC article.
-
Virus-like particles of the Ty3 retrotransposon assemble in association with P-body components.RNA. 2006 Jan;12(1):94-101. doi: 10.1261/rna.2264806. RNA. 2006. PMID: 16373495 Free PMC article.
-
Ty3, a Position-specific Retrotransposon in Budding Yeast.Microbiol Spectr. 2015 Apr;3(2):MDNA3-0057-2014. doi: 10.1128/microbiolspec.MDNA3-0057-2014. Microbiol Spectr. 2015. PMID: 26104707 Review.
-
Function of a retrotransposon nucleocapsid protein.RNA Biol. 2010 Nov-Dec;7(6):642-54. doi: 10.4161/rna.7.6.14117. Epub 2010 Nov 1. RNA Biol. 2010. PMID: 21189452 Free PMC article. Review.
Cited by
-
XRN1 Is a Species-Specific Virus Restriction Factor in Yeasts.PLoS Pathog. 2016 Oct 6;12(10):e1005890. doi: 10.1371/journal.ppat.1005890. eCollection 2016 Oct. PLoS Pathog. 2016. PMID: 27711183 Free PMC article.
-
A prion-like domain in Gag capsid protein drives retrotransposon particle assembly and mobility.Proc Natl Acad Sci U S A. 2023 Aug 29;120(35):e2311419120. doi: 10.1073/pnas.2311419120. Epub 2023 Aug 17. Proc Natl Acad Sci U S A. 2023. PMID: 37590432 Free PMC article. No abstract available.
-
A self-encoded capsid derivative restricts Ty1 retrotransposition in Saccharomyces.Curr Genet. 2016 May;62(2):321-9. doi: 10.1007/s00294-015-0550-6. Epub 2015 Dec 9. Curr Genet. 2016. PMID: 26650614 Free PMC article. Review.
-
Structure of the Ty3/Gypsy retrotransposon capsid and the evolution of retroviruses.Proc Natl Acad Sci U S A. 2019 May 14;116(20):10048-10057. doi: 10.1073/pnas.1900931116. Epub 2019 Apr 29. Proc Natl Acad Sci U S A. 2019. PMID: 31036670 Free PMC article.
-
Ribonucleoprotein Granules: Between Stress and Transposable Elements.Biomolecules. 2023 Jun 23;13(7):1027. doi: 10.3390/biom13071027. Biomolecules. 2023. PMID: 37509063 Free PMC article. Review.
References
-
- Eulalio A, Behm-Ansmant I, Izaurralde E. P bodies: at the crossroads of post-transcriptional pathways. Nat Rev Mol Cell Biol. 2007;8(1):9–22. Epub 2006/12/22. nrm2080 [pii] 0.1038/nrm2080. . - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

