Sparing of the extraocular muscles in mdx mice with absent or reduced utrophin expression: A life span analysis
- PMID: 26429098
- PMCID: PMC4630113
- DOI: 10.1016/j.nmd.2015.09.001
Sparing of the extraocular muscles in mdx mice with absent or reduced utrophin expression: A life span analysis
Abstract
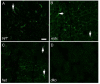
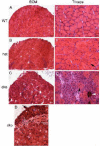
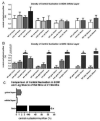
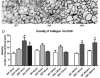
Sparing of the extraocular muscles in muscular dystrophy is controversial. To address the potential role of utrophin in this sparing, mdx:utrophin(+/-) and mdx:utrophin(-/-) mice were examined for changes in myofiber size, central nucleation, and Pax7-positive and MyoD-positive cell density at intervals over their life span. Known to be spared in the mdx mouse, and contrary to previous reports, the extraocular muscles from both the mdx:utrophin(+/-) and mdx:utrophin(-/-) mice were also morphologically spared. In the mdx:utrophin(+/)(-) mice, which have a normal life span compared to the mdx:utrophin(-/-) mice, the myofibers were larger at 3 and 12 months than the wild type age-matched eye muscles. While there was a significant increase in central nucleation in the extraocular muscles from all mdx:utrophin(+/)(-) mice, the levels were still very low compared to age-matched limb skeletal muscles. Pax7- and MyoD-positive myogenic precursor cell populations were retained and were similar to age-matched wild type controls. These results support the hypothesis that utrophin is not involved in extraocular muscle sparing in these genotypes. In addition, it appears that these muscles retain the myogenic precursors that would allow them to maintain their regenerative capacity and normal morphology over a lifetime even in these more severe models of muscular dystrophy.
Keywords: Extraocular muscle; Muscular dystrophy; MyoD; Pax7; Satellite cell; Utrophin.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures




Similar articles
-
Dystrophic changes in extraocular muscles after gamma irradiation in mdx:utrophin(+/-) mice.PLoS One. 2014 Jan 21;9(1):e86424. doi: 10.1371/journal.pone.0086424. eCollection 2014. PLoS One. 2014. PMID: 24466085 Free PMC article.
-
The sparing of extraocular muscle in dystrophinopathy is lost in mice lacking utrophin and dystrophin.J Cell Sci. 1998 Jul;111 ( Pt 13):1801-11. doi: 10.1242/jcs.111.13.1801. J Cell Sci. 1998. PMID: 9625743
-
Satellite cells and utrophin are not directly correlated with the degree of skeletal muscle damage in mdx mice.Am J Physiol Cell Physiol. 2005 Jul;289(1):C42-8. doi: 10.1152/ajpcell.00577.2004. Epub 2005 Feb 9. Am J Physiol Cell Physiol. 2005. PMID: 15703201
-
Analysis of gene expression differences between utrophin/dystrophin-deficient vs mdx skeletal muscles reveals a specific upregulation of slow muscle genes in limb muscles.Neurogenetics. 2006 May;7(2):81-91. doi: 10.1007/s10048-006-0031-7. Epub 2006 Mar 9. Neurogenetics. 2006. PMID: 16525850
-
Eye muscle sparing by the muscular dystrophies: lessons to be learned?Microsc Res Tech. 2000 Feb 1-15;48(3-4):192-203. doi: 10.1002/(SICI)1097-0029(20000201/15)48:3/4<192::AID-JEMT7>3.0.CO;2-J. Microsc Res Tech. 2000. PMID: 10679966 Review.
Cited by
-
Skeletal muscle in health and disease.Dis Model Mech. 2020 Feb 6;13(2):dmm042192. doi: 10.1242/dmm.042192. Dis Model Mech. 2020. PMID: 32066552 Free PMC article. Review.
-
Mass Spectrometric Profiling of Extraocular Muscle and Proteomic Adaptations in the mdx-4cv Model of Duchenne Muscular Dystrophy.Life (Basel). 2021 Jun 22;11(7):595. doi: 10.3390/life11070595. Life (Basel). 2021. PMID: 34206383 Free PMC article.
References
-
- Marques MJ, Ferretti R, Vomero VU, Minatel E, Neto HS. Intrinsic laryngeal muscles are spared from myonecrosis in the mdx mouse model of Duchenne muscular dystrophy. Muscle Nerve. 2007;35:349–353. - PubMed
-
- Vereecken RL, Verduyn H. The electrical activity of the paraurethral and perineal muscles in normal and pathological conditions. Br. J. Urol. 1970;2:457–463. - PubMed
-
- Karpati G, Carpenter S. Small caliber skeletal muscle fibers do not suffer deleterious consequences of dystrophic gene expression. Am. J. Med. Genet. 1986;25:653–658. - PubMed
-
- Kaminski HJ, al-Hakim M, Leigh RJ, Katirji MB, Ruff RL. Extraocular muscles are spared in advanced Duchenne dystrophy. Ann. Neurol. 1992;32:586–588. - PubMed
-
- McLoon LK. In: The Extraocular Muscles. In: Adler's Physiology of the Eye. Kaufman P, Alm A, Levin LA, Nilsson S, Ver Hoeve J, Wu SM, editors. Mosby Press; Missouri: 2011. pp. 182–207.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

