N-acetylcysteine reverses diastolic dysfunction and hypertrophy in familial hypertrophic cardiomyopathy
- PMID: 26432840
- PMCID: PMC4666985
- DOI: 10.1152/ajpheart.00339.2015
N-acetylcysteine reverses diastolic dysfunction and hypertrophy in familial hypertrophic cardiomyopathy
Abstract
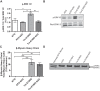
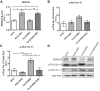
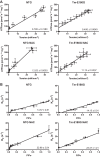
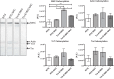
S-glutathionylation of cardiac myosin-binding protein C (cMyBP-C) induces Ca(2+) sensitization and a slowing of cross-bridge kinetics as a result of increased oxidative signaling. Although there is evidence for a role of oxidative stress in disorders associated with hypertrophic cardiomyopathy (HCM), this mechanism is not well understood. We investigated whether oxidative myofilament modifications may be in part responsible for diastolic dysfunction in HCM. We administered N-acetylcysteine (NAC) for 30 days to 1-mo-old wild-type mice and to transgenic mice expressing a mutant tropomyosin (Tm-E180G) and nontransgenic littermates. Tm-E180G hearts demonstrate a phenotype similar to human HCM. After NAC administration, the morphology and diastolic function of Tm-E180G mice was not significantly different from controls, indicating that NAC had reversed baseline diastolic dysfunction and hypertrophy in our model. NAC administration also increased sarco(endo)plasmic reticulum Ca(2+) ATPase protein expression, reduced extracellular signal-related kinase 1/2 phosphorylation, and normalized phosphorylation of phospholamban, as assessed by Western blot. Detergent-extracted fiber bundles from NAC-administered Tm-E180G mice showed nearly nontransgenic (NTG) myofilament Ca(2+) sensitivity. Additionally, we found that NAC increased tension cost and rate of cross-bridge reattachment. Tm-E180G myofilaments were found to have a significant increase in S-glutathionylation of cMyBP-C, which was returned to NTG levels upon NAC administration. Taken together, our results indicate that oxidative myofilament modifications are an important mediator in diastolic function, and by relieving this modification we were able to reverse established diastolic dysfunction and hypertrophy in HCM.
Keywords: S-glutathionylation; cardiac myosin-binding protein C; diastolic dysfunction; oxidative stress; sarcomeres.
Copyright © 2015 the American Physiological Society.
Figures



References
-
- Alves ML, Dias FA, Gaffin RD, Simon JN, Montminy EM, Biesiadecki BJ, Hinken AC, Warren CM, Utter MS, Davis RT 3rd, Sadayappan S, Robbins J, Wieczorek DF, Solaro RJ, Wolska BM. Desensitization of myofilaments to Ca2+ as a therapeutic target for hypertrophic cardiomyopathy with mutations in thin filament proteins. Circ Cardiovasc Genet 7: 132–143, 2014. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

