Characterization of cytopathogenicity of classical swine fever virus isolate induced by Newcastle disease virus
- PMID: 26436124
- PMCID: PMC4585056
- DOI: 10.1007/s13337-015-0253-0
Characterization of cytopathogenicity of classical swine fever virus isolate induced by Newcastle disease virus
Abstract
Classical swine fever virus (CSFV), the causative agent of classical swine fever, belongs to the family Flaviviridae and genus Pestivirus. Some pestiviruses exhibit cytopathic effect in cell culture but exact phenomenon is unknown. Over expression of NS2-3 gene, presence of defective interfering particle and exaltation of Newcastle disease virus (END) phenomenon could be the reasons of cytopathogenicity. In the present study, a CSFV isolate exhibiting cytopathic effect (CPE) in Madin-Darby Canine Kidney (MDCK) cell line was characterized. To characterize cytopathogenicity of such isolate, END test was carried out. Interference of Newcastle disease virus (NDV) in MDCK adapted CSFV was confirmed by RT-PCR and virus neutralization test. Absence of CPE and NDV specific nucleic acid after neutralization confirmed the induction of CPE by NDV. Further, identity of the CSFV isolate in MDCK cell line by immunoperoxidase test, immunoblotting and RT-PCR post NDV neutralization established the virus replication without CPE (non-cytopathic isolate). Findings suggest that, there could be a chance of mixed infection of both CSFV and NDV in the piglet from which the sample was collected for virus isolation.
Keywords: CSF; Cytopathogenicity; END phenomenon; MDCK; VNT; Virus.
Figures

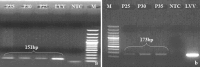

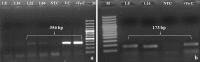

References
-
- Aoki H, Ishikawa K, Sakoda Y, Sekiguchi H, Kodama M, Suzuki S, Fukusho A. Characterization of classical swine fever virus associated with defective interfering particles containing a cytopathogenic subgenomic RNA isolated from wild boar. J Vet Med Sci. 2001;63:751–758. doi: 10.1292/jvms.63.751. - DOI - PubMed
-
- Cheng LF, Ke MF, Zheng T, Huang Y, Li WY, Shi SH. Isolation and identification of Newcastle disease virus from swine. Chin J Vet Sci Tech. 2004–12. http://en.cnki.com.cn/Article_en/CJFDTOTAL-ZGSY200412020.htm.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

