Synaptotagmin-1 and -7 Are Redundantly Essential for Maintaining the Capacity of the Readily-Releasable Pool of Synaptic Vesicles
- PMID: 26437117
- PMCID: PMC4593569
- DOI: 10.1371/journal.pbio.1002267
Synaptotagmin-1 and -7 Are Redundantly Essential for Maintaining the Capacity of the Readily-Releasable Pool of Synaptic Vesicles
Abstract
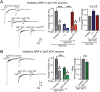
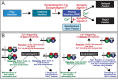
In forebrain neurons, Ca(2+) triggers exocytosis of readily releasable vesicles by binding to synaptotagmin-1 and -7, thereby inducing fast and slow vesicle exocytosis, respectively. Loss-of-function of synaptotagmin-1 or -7 selectively impairs the fast and slow phase of release, respectively, but does not change the size of the readily-releasable pool (RRP) of vesicles as measured by stimulation of release with hypertonic sucrose, or alter the rate of vesicle priming into the RRP. Here we show, however, that simultaneous loss-of-function of both synaptotagmin-1 and -7 dramatically decreased the capacity of the RRP, again without altering the rate of vesicle priming into the RRP. Either synaptotagmin-1 or -7 was sufficient to rescue the RRP size in neurons lacking both synaptotagmin-1 and -7. Although maintenance of RRP size was Ca(2+)-independent, mutations in Ca(2+)-binding sequences of synaptotagmin-1 or synaptotagmin-7--which are contained in flexible top-loop sequences of their C2 domains--blocked the ability of these synaptotagmins to maintain the RRP size. Both synaptotagmins bound to SNARE complexes; SNARE complex binding was reduced by the top-loop mutations that impaired RRP maintenance. Thus, synaptotagmin-1 and -7 perform redundant functions in maintaining the capacity of the RRP in addition to nonredundant functions in the Ca(2+) triggering of different phases of release.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

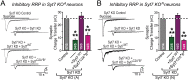

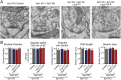
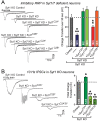

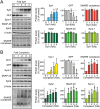

Comment in
-
Dual-Function Exocytosis Regulator Has Yet Another Job.PLoS Biol. 2015 Oct 5;13(10):e1002268. doi: 10.1371/journal.pbio.1002268. eCollection 2015 Oct. PLoS Biol. 2015. PMID: 26437253 Free PMC article.
References
-
- Sabatini BL, Regehr WG. Timing of neurotransmission at fast synapses in the mammalian brain. Nature. 1996;384(6605):170–2. Epub 1996/11/14. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

