A Large-Scale Circuit Mechanism for Hierarchical Dynamical Processing in the Primate Cortex
- PMID: 26439530
- PMCID: PMC4630024
- DOI: 10.1016/j.neuron.2015.09.008
A Large-Scale Circuit Mechanism for Hierarchical Dynamical Processing in the Primate Cortex
Abstract
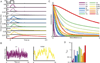
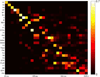
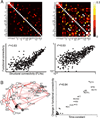
We developed a large-scale dynamical model of the macaque neocortex, which is based on recently acquired directed- and weighted-connectivity data from tract-tracing experiments, and which incorporates heterogeneity across areas. A hierarchy of timescales naturally emerges from this system: sensory areas show brief, transient responses to input (appropriate for sensory processing), whereas association areas integrate inputs over time and exhibit persistent activity (suitable for decision-making and working memory). The model displays multiple temporal hierarchies, as evidenced by contrasting responses to visual versus somatosensory stimulation. Moreover, slower prefrontal and temporal areas have a disproportionate impact on global brain dynamics. These findings establish a circuit mechanism for "temporal receptive windows" that are progressively enlarged along the cortical hierarchy, suggest an extension of time integration in decision making from local to large circuits, and should prompt a re-evaluation of the analysis of functional connectivity (measured by fMRI or electroencephalography/magnetoencephalography) by taking into account inter-areal heterogeneity.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures





Comment in
-
Processing Timescales as an Organizing Principle for Primate Cortex.Neuron. 2015 Oct 21;88(2):244-6. doi: 10.1016/j.neuron.2015.10.010. Neuron. 2015. PMID: 26494274
References
-
- Abbott LF, Chance FS. Drivers and modulators from push-pull and balanced synaptic input. Prog. Brain Res. 2005;149:147–155. - PubMed
-
- Amit DJ, Fusi S, Yakovlev V. Paradigmatic working memory (attractor) cell in IT cortex. Neural Comput. 1997;9:1071–1092. - PubMed
-
- Barbas H, Rempel-Clower N. Cortical structure predicts the pattern of corticocortical connections. Cereb. Cortex. 1997;7:635–646. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

