S-1 plus cisplatin versus fluorouracil plus cisplatin in advanced gastric or gastro-esophageal junction adenocarcinoma patients: a pilot study
- PMID: 26439700
- PMCID: PMC4741513
- DOI: 10.18632/oncotarget.5959
S-1 plus cisplatin versus fluorouracil plus cisplatin in advanced gastric or gastro-esophageal junction adenocarcinoma patients: a pilot study
Abstract
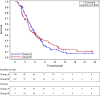
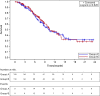
The safety and efficacy of S-1 plus cisplatin in Chinese advanced gastric cancer patients in first line setting is unknown. In this pilot study, patients with advanced gastric or gastro-esophageal junction adenocarcinoma were enrolled and randomly assigned in a 1:1 ratio to receive S-1 plus cisplatin (CS group) or 5-FU plus cisplatin (CF group). The primary endpoint was time to progression (TTP). Secondary end points included overall survival (OS) and safety. This study was registered on ClinicalTrials. Gov, number NCT01198392. A total of 236 patients were enrolled. Median TTP was 5.51 months in CS group compared with 4.62 months in CF group [hazard ratio (HR) 1.028, 95% confidential interval (CI) 0.758-1.394, p = 0.859]. Median OS was 10.00 months and 10.46 months in CS and CF groups (HR 1.046, 95%CI 0.709-1.543, p = 0.820), respectively. The most common adverse events in both groups were anemia, leukopenia, neutropenia, nausea, thrombocytopenia, vomiting, anorexia and diarrhea. We find that S-1 plus cisplatin is an effective and tolerable option for advanced gastric or gastro-esophageal junction adenocarcinoma patients in China.
Keywords: S-1; first-line chemotherapy; fluorouracil; gastric cancer.
Conflict of interest statement
All authors declared that there is no conflict of interest.
Figures
References
-
- Moore MA, Eser S, Igisinov N, Igisinov S, Mohagheghi MA, Mousavi-Jarrahi A, Ozenturk G, Soipova M, Tuncer M, Sobue T. Cancer epidemiology and control in North-Western and Central Asia - past, present and future. Asian Pac J Cancer Prev. 2010;(11 Suppl 2):17–32. - PubMed
-
- Qiu MZ, Wang ZQ, Zhang DS, Liu Q, Luo HY, Zhou ZW, Li YH, Jiang WQ, Xu RH. Comparison of 6th and 7th AJCC TNM staging classification for carcinoma of the stomach in China. Ann Surg Oncol. 2011;18:1869–1876. - PubMed
-
- Cullinan SA, Moertel CG, Fleming TR, Rubin JR, Krook JE, Everson LK, Windschitl HE, Twito DI, Marschke RF, Foley JF, et al. A comparison of three chemotherapeutic regimens in the treatment of advanced pancreatic and gastric carcinoma. Fluorouracil vs fluorouracil and doxorubicin vs fluorouracil, doxorubicin, and mitomycin. Jama. 1985;253:2061–2067. - PubMed
-
- Cunningham D, Starling N, Rao S, Iveson T, Nicolson M, Coxon F, Middleton G, Daniel F, Oates J, Norman AR. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med. 2008;358:36–46. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical