Ion-pumping microbial rhodopsins
- PMID: 26442282
- PMCID: PMC4585134
- DOI: 10.3389/fmolb.2015.00052
Ion-pumping microbial rhodopsins
Abstract
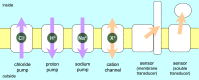
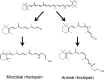
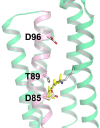
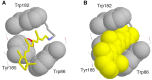
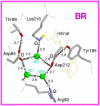
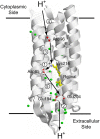
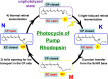
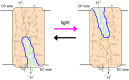
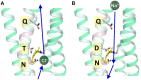
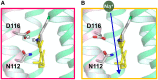
Rhodopsins are light-sensing proteins used in optogenetics. The word "rhodopsin" originates from the Greek words "rhodo" and "opsis," indicating rose and sight, respectively. Although the classical meaning of rhodopsin is the red-colored pigment in our eyes, the modern meaning of rhodopsin encompasses photoactive proteins containing a retinal chromophore in animals and microbes. Animal and microbial rhodopsins possess 11-cis and all-trans retinal, respectively, to capture light in seven transmembrane α-helices, and photoisomerizations into all-trans and 13-cis forms, respectively, initiate each function. Ion-transporting proteins can be found in microbial rhodopsins, such as light-gated channels and light-driven pumps, which are the main tools in optogenetics. Light-driven pumps, such as archaeal H(+) pump bacteriorhodopsin (BR) and Cl(-) pump halorhodopsin (HR), were discovered in the 1970s, and their mechanism has been extensively studied. On the other hand, different kinds of H(+) and Cl(-) pumps have been found in marine bacteria, such as proteorhodopsin (PR) and Fulvimarina pelagi rhodopsin (FR), respectively. In addition, a light-driven Na(+) pump was found, Krokinobacter eikastus rhodopsin 2 (KR2). These light-driven ion-pumping microbial rhodopsins are classified as DTD, TSA, DTE, NTQ, and NDQ rhodopsins for BR, HR, PR, FR, and KR2, respectively. Recent understanding of ion-pumping microbial rhodopsins is reviewed in this paper.
Keywords: H+ transfer; hydrogen bond; light-driven pump; photocycle; photoisomerizatoin; retinal; structural change.
Figures


References
-
- Agmon N. (1995). The Grotthuss mechanism. Chem. Phys. Lett. 244, 456–462. 10.1016/0009-2614(95)00905-J - DOI
-
- Balashov S. P., Petrovskaya L. E., Imasheva E. S., Lukashev E. P., Dioumaev A. K., Wang J. M., et al. (2013). Breaking the carboxyl rule. Lysine96 facilitates protonation of the Schifff base in the photocycle of a retinal protein from Exiguobacterium sibiricum. J. Biol. Chem. 288, 21254–21265. 10.1074/jbc.M113.465138 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

