APOE Isoforms Control Pathogenic Subretinal Inflammation in Age-Related Macular Degeneration
- PMID: 26446211
- PMCID: PMC6605380
- DOI: 10.1523/JNEUROSCI.2468-15.2015
APOE Isoforms Control Pathogenic Subretinal Inflammation in Age-Related Macular Degeneration
Abstract
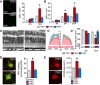
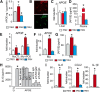
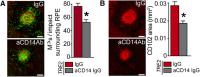
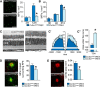
Contrary to Alzheimer's disease (AD), the APOE2 allele increases and the APOE4 allele reduces the risk to develop age-related macular degeneration (AMD) compared with the most common APOE3 allele. The underlying mechanism for this association with AMD and the reason for the puzzling difference with AD are unknown. We previously demonstrated that pathogenic subretinal mononuclear phagocytes (MPs) accumulate in Cx3cr1-deficient mice due to the overexpression of APOE, interleukin-6, and CC chemokine ligand 2 (CCL2). We here show using targeted replacement mice expressing the human APOE isoforms (TRE2, TRE3, and TRE4) that MPs of TRE2 mice express increased levels of APOE, interleukin-6, and CCL2 and develop subretinal MP accumulation, photoreceptor degeneration, and exaggerated choroidal neovascularization similar to AMD. Pharmacological inhibition of the cytokine induction inhibited the pathogenic subretinal inflammation. In the context of APOE-dependent subretinal inflammation in Cx3cr1(GFP/GFP) mice, the APOE4 allele led to diminished APOE and CCL2 levels and protected Cx3cr1(GFP/GFP) mice against harmful subretinal MP accumulation observed in Cx3cr1(GFP/GFP)TRE3 mice. Our study shows that pathogenic subretinal inflammation is APOE isoform-dependent and provides the rationale for the previously unexplained implication of the APOE2 isoform as a risk factor and the APOE4 isoform as a protective factor in AMD pathogenesis.
Significance statement: The understanding of how genetic predisposing factors, which play a major role in age-related macular degeneration (AMD), participate in its pathogenesis is an important clue to decipher the pathomechanism and develop efficient therapies. In this study, we used transgenic, targeted replacement mice that carry the three human APOE isoform-defining sequences at the mouse APOE chromosomal location and express the human APOE isoforms. Our study is the first to show how APOE2 provokes and APOE4 inhibits the cardinal AMD features, inflammation, degeneration, and exaggerated neovascularization. Our findings reflect the clinical association of the genetic predisposition that was recently confirmed in a major pooled analysis. They emphasize the role of APOE in inflammation and inflammation in AMD.
Keywords: apolipoprotein E; mononuclear phagocyte; neurodegeneration; neuroinflammation.
Copyright © 2015 the authors 0270-6474/15/3513568-09$15.00/0.
Figures
References
-
- Anderson DH, Ozaki S, Nealon M, Neitz J, Mullins RF, Hageman GS, Johnson LV. Local cellular sources of apolipoprotein E in the human retina and retinal pigmented epithelium: implications for the process of drusen formation. Am J Ophthalmol. 2001;131:767–781. doi: 10.1016/S0002-9394(00)00961-2. - DOI - PubMed
-
- Chen W, Stambolian D, Edwards AO, Branham KE, Othman M, Jakobsdottir J, Tosakulwong N, Pericak-Vance MA, Campochiaro PA, Klein ML, Tan PL, Conley YP, Kanda A, Kopplin L, Li Y, Augustaitis KJ, Karoukis AJ, Scott WK, Agarwal A, Kovach JL, et al. Genetic variants near TIMP3 and high-density lipoprotein-associated loci influence susceptibility to age-related macular degeneration. Proc Natl Acad Sci U S A. 2010;107:7401–7406. doi: 10.1073/pnas.0912702107. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous
