Downregulation of Sphingosine 1-Phosphate Receptor 1 Promotes the Switch from Tangential to Radial Migration in the OB
- PMID: 26446219
- PMCID: PMC6605378
- DOI: 10.1523/JNEUROSCI.1353-15.2015
Downregulation of Sphingosine 1-Phosphate Receptor 1 Promotes the Switch from Tangential to Radial Migration in the OB
Abstract
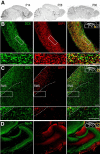
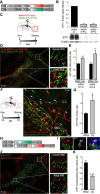
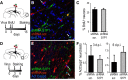
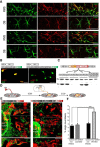
Neuroblast migration is a highly orchestrated process that ensures the proper integration of newborn neurons into complex neuronal circuits. In the postnatal rodent brain, neuroblasts migrate long distances from the subependymal zone of the lateral ventricles to the olfactory bulb (OB) within the rostral migratory stream (RMS). They first migrate tangentially in close contact to each other and later radially as single cells until they reach their final destination in the OB. Sphingosine 1-phosphate (S1P) is a bioactive lipid that interacts with cell-surface receptors to exert different cellular responses. Although well studied in other systems and a target for the treatment of multiple sclerosis, little is known about S1P in the postnatal brain. Here, we report that the S1P receptor 1 (S1P1) is expressed in neuroblasts migrating in the RMS. Using in vivo and in vitro gain- and loss-of-function approaches in both wild-type and transgenic mice, we found that the activation of S1P1 by its natural ligand S1P, acting as a paracrine signal, contributes to maintain neuroblasts attached to each other while they migrate in chains within the RMS. Once in the OB, neuroblasts cease to express S1P1, which results in cell detachment and initiation of radial migration, likely via downregulation of NCAM1 and β1 integrin. Our results reveal a novel physiological function for S1P1 in the postnatal brain, directing the path followed by newborn neurons in the neurogenic niche.
Significance statement: The function of each neuron is highly determined by the position it occupies within a neuronal circuit. Frequently, newborn neurons must travel long distances from their birthplace to their predetermined final location and, to do so, they use different modes of migration. In this study, we identify the sphingosine 1-phosphate (S1P) receptor 1 (S1P1) as one of the key players that govern the switch from tangential to radial migration of postnatally generated neuroblasts in the olfactory bulb. Of interest is the evidence that the ligand, S1P, is provided by nearby astrocytes. Finally, we also propose adhesion molecules that act downstream of S1P1 and initiate the transition from tangential chain migration to individual radial migration outside of the stream.
Keywords: cell adhesion; migration; olfactory bulb; retrovirus; sphingosine; time lapse.
Copyright © 2015 the authors 0270-6474/15/3513659-14$15.00/0.
Figures




References
-
- Bayer SA. 3H-thymidine-radiographic studies of neurogenesis in the rat olfactory bulb. Exp Brain Res. 1983;50:329–340. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
