MiRNA-891a-5p mediates HIV-1 Tat and KSHV Orf-K1 synergistic induction of angiogenesis by activating NF-κB signaling
- PMID: 26446987
- PMCID: PMC4627096
- DOI: 10.1093/nar/gkv988
MiRNA-891a-5p mediates HIV-1 Tat and KSHV Orf-K1 synergistic induction of angiogenesis by activating NF-κB signaling
Erratum in
-
MiRNA-891a-5p mediates HIV-1 Tat and KSHV Orf-K1 synergistic induction of angiogenesis by activating NF-κB signaling.Nucleic Acids Res. 2019 Mar 18;47(5):2700. doi: 10.1093/nar/gkz088. Nucleic Acids Res. 2019. PMID: 30726947 Free PMC article. No abstract available.
Abstract
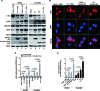
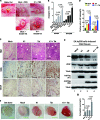
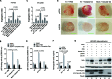
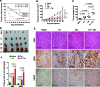
Co-infection with HIV-1 and Kaposi's sarcoma-associated herpesvirus (KSHV) is the cause of aggressive AIDS-related Kaposi's sarcoma (AIDS-KS) characterized by abnormal angiogenesis. The impact of HIV-1 and KSHV interaction on the pathogenesis and extensive angiogenesis of AIDS-KS remains unclear. Here, we explored the synergistic effect of HIV-1 Tat and KSHV oncogene Orf-K1 on angiogenesis. Our results showed that soluble Tat or ectopic expression of Tat enhanced K1-induced cell proliferation, microtubule formation and angiogenesis in chorioallantoic membrane and nude mice models. Mechanistic studies revealed that Tat promoted K1-induced angiogenesis by enhancing NF-κB signaling. Mechanistically, we showed that Tat synergized with K1 to induce the expression of miR-891a-5p, which directly targeted IκBα 3' untranslated region, leading to NF-κB activation. Consequently, inhibition of miR-891a-5p increased IκBα level, prevented nuclear translocation of NF-κB p65 and ultimately suppressed the synergistic effect of Tat- and K1-induced angiogenesis. Our results illustrate that, by targeting IκBα to activate the NF-κB pathway, miR-891a-5p mediates Tat and K1 synergistic induction of angiogenesis. Therefore, the miR-891a-5p/NF-κB pathway is important in the pathogenesis of AIDS-KS, which could be an attractive therapeutic target for AIDS-KS.
© The Author(s) 2015. Published by Oxford University Press on behalf of Nucleic Acids Research.
Figures





Similar articles
-
HIV-1 Tat-induced disruption of epithelial junctions and epithelial-mesenchymal transition of oral and genital epithelial cells lead to increased invasiveness of neoplastic cells and the spread of herpes simplex virus and cytomegalovirus.Front Immunol. 2025 Feb 13;16:1541532. doi: 10.3389/fimmu.2025.1541532. eCollection 2025. Front Immunol. 2025. PMID: 40018040 Free PMC article. Review.
-
HIV-1 Vpr Inhibits Kaposi's Sarcoma-Associated Herpesvirus Lytic Replication by Inducing MicroRNA miR-942-5p and Activating NF-κB Signaling.J Virol. 2016 Sep 12;90(19):8739-53. doi: 10.1128/JVI.00797-16. Print 2016 Oct 1. J Virol. 2016. PMID: 27440900 Free PMC article.
-
Upregulation of MicroRNA 711 Mediates HIV-1 Vpr Promotion of Kaposi's Sarcoma-Associated Herpesvirus Latency and Induction of Pro-proliferation and Pro-survival Cytokines by Targeting the Notch/NF-κB-Signaling Axis.J Virol. 2018 Aug 29;92(18):e00580-18. doi: 10.1128/JVI.00580-18. Print 2018 Sep 15. J Virol. 2018. PMID: 29976660 Free PMC article.
-
HIV-1 Nef and KSHV oncogene K1 synergistically promote angiogenesis by inducing cellular miR-718 to regulate the PTEN/AKT/mTOR signaling pathway.Nucleic Acids Res. 2014 Sep;42(15):9862-79. doi: 10.1093/nar/gku583. Epub 2014 Aug 7. Nucleic Acids Res. 2014. PMID: 25104021 Free PMC article.
-
Regulation of the MIR155 host gene in physiological and pathological processes.Gene. 2013 Dec 10;532(1):1-12. doi: 10.1016/j.gene.2012.12.009. Epub 2012 Dec 14. Gene. 2013. PMID: 23246696 Review.
Cited by
-
The SH3BGR/STAT3 Pathway Regulates Cell Migration and Angiogenesis Induced by a Gammaherpesvirus MicroRNA.PLoS Pathog. 2016 Apr 29;12(4):e1005605. doi: 10.1371/journal.ppat.1005605. eCollection 2016 Apr. PLoS Pathog. 2016. PMID: 27128969 Free PMC article.
-
MicroRNAs in the Vitreous Humor of Patients with Retinal Detachment and a Different Grading of Proliferative Vitreoretinopathy: A Pilot Study.Transl Vis Sci Technol. 2020 May 22;9(6):23. doi: 10.1167/tvst.9.6.23. eCollection 2020 May. Transl Vis Sci Technol. 2020. PMID: 32821520 Free PMC article.
-
CircRNA ARFGEF1 functions as a ceRNA to promote oncogenic KSHV-encoded viral interferon regulatory factor induction of cell invasion and angiogenesis by upregulating glutaredoxin 3.PLoS Pathog. 2021 Feb 4;17(2):e1009294. doi: 10.1371/journal.ppat.1009294. eCollection 2021 Feb. PLoS Pathog. 2021. PMID: 33539420 Free PMC article.
-
HIV-1 Tat-induced disruption of epithelial junctions and epithelial-mesenchymal transition of oral and genital epithelial cells lead to increased invasiveness of neoplastic cells and the spread of herpes simplex virus and cytomegalovirus.Front Immunol. 2025 Feb 13;16:1541532. doi: 10.3389/fimmu.2025.1541532. eCollection 2025. Front Immunol. 2025. PMID: 40018040 Free PMC article. Review.
-
Human immunodeficiency virus Tat-TIP30 interaction promotes metastasis by enhancing the nuclear translocation of Snail in lung cancer cell lines.Cancer Sci. 2018 Oct;109(10):3105-3114. doi: 10.1111/cas.13768. Epub 2018 Sep 3. Cancer Sci. 2018. PMID: 30099830 Free PMC article.
References
-
- Chang Y., Cesarman E., Pessin M.S., Lee F., Culpepper J., Knowles D.M., Moore P.S. Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi's sarcoma. Science. 1994;266:1865–1869. - PubMed
-
- Ganem D. KSHV infection and the pathogenesis of Kaposi's sarcoma. Annu. Rev. Pathol. 2006;1:273–296. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

