Neural crest-mediated bone resorption is a determinant of species-specific jaw length
- PMID: 26449912
- PMCID: PMC4698309
- DOI: 10.1016/j.ydbio.2015.10.001
Neural crest-mediated bone resorption is a determinant of species-specific jaw length
Abstract
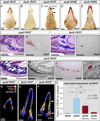
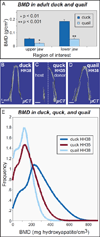
Precise control of jaw length during development is crucial for proper form and function. Previously we have shown that in birds, neural crest mesenchyme (NCM) confers species-specific size and shape to the beak by regulating molecular and histological programs for the induction and deposition of cartilage and bone. Here we reveal that a hitherto unrecognized but similarly essential mechanism for establishing jaw length is the ability of NCM to mediate bone resorption. Osteoclasts are considered the predominant cells that resorb bone, although osteocytes have also been shown to participate in this process. In adults, bone resorption is tightly coupled to bone deposition as a means to maintain skeletal homeostasis. Yet, the role and regulation of bone resorption during growth of the embryonic skeleton have remained relatively unexplored. We compare jaw development in short-beaked quail versus long-billed duck and find that quail have substantially higher levels of enzymes expressed by bone-resorbing cells including tartrate-resistant acid phosphatase (TRAP), Matrix metalloproteinase 13 (Mmp13), and Mmp9. Then, we transplant NCM destined to form the jaw skeleton from quail to duck and generate chimeras in which osteocytes arise from quail donor NCM and osteoclasts come exclusively from the duck host. Chimeras develop quail-like jaw skeletons coincident with dramatically elevated expression of TRAP, Mmp13, and Mmp9. To test for a link between bone resorption and jaw length, we block resorption using a bisphosphonate, osteoprotegerin protein, or an MMP13 inhibitor, and this significantly lengthens the jaw. Conversely, activating resorption with RANKL protein shortens the jaw. Finally, we find that higher resorption in quail presages their relatively lower adult jaw bone mineral density (BMD) and that BMD is also NCM-mediated. Thus, our experiments suggest that NCM not only controls bone resorption by its own derivatives but also modulates the activity of mesoderm-derived osteoclasts, and in so doing enlists bone resorption as a key patterning mechanism underlying the functional morphology and evolution of the jaw.
Keywords: Avian beak development and evolution; Bisphosphonates; Bone mineral density; Bone remodeling; Bone resorption; Cranial neural crest; Evolutionary developmental biology; Jaw length; Mmp13; Mmp9; OPG; Osteoclasts; Osteocytes; Osteocytic osteolysis; Quail-duck chimeras; Quck; RANKL; TRAP staining.
Copyright © 2015 The Authors. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Abzhanov A, Kuo WP, Hartmann C, Grant BR, Grant PR, Tabin CJ. The calmodulin pathway and evolution of elongated beak morphology in Darwin’s finches. Nature. 2006;442:563–567. - PubMed
-
- Abzhanov A, Protas M, Grant BR, Grant PR, Tabin CJ. Bmp4 and morphological variation of beaks in Darwin’s finches. Science. 2004;305:1462–1465. - PubMed
-
- Abzhanov A, Tabin CJ. Shh and Fgf8 act synergistically to drive cartilage outgrowth during cranial development. Dev. Biol. 2004;273:134–148. - PubMed
-
- Albrecht UEG, Helms JA, Lin H. Visualization of gene expression patterns by in situ hybridization. In: Daston GP, editor. Molecular and Cellular Methods in Developmental Toxicology. Boca Raton, FL: CRC Press; 1997. pp. 23–48.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

