PI3K/AKT/mTOR and sonic hedgehog pathways cooperate together to inhibit human pancreatic cancer stem cell characteristics and tumor growth
- PMID: 26451606
- PMCID: PMC4741658
- DOI: 10.18632/oncotarget.5055
PI3K/AKT/mTOR and sonic hedgehog pathways cooperate together to inhibit human pancreatic cancer stem cell characteristics and tumor growth
Abstract
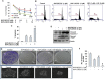
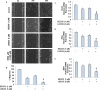
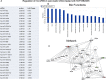
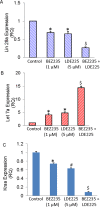
Cancer stem cells (CSCs) play major roles in cancer initiation, progression, and metastasis. It is evident from growing reports that PI3K/Akt/mTOR and Sonic Hedgehog (Shh) signaling pathways are aberrantly reactivated in pancreatic CSCs. Here, we examined the efficacy of combining NVP-LDE-225 (PI3K/mTOR inhibitor) and NVP-BEZ-235 (Smoothened inhibitor) on pancreatic CSCs characteristics, microRNA regulatory network, and tumor growth. NVP-LDE-225 co-operated with NVP-BEZ-235 in inhibiting pancreatic CSC's characteristics and tumor growth in mice by acting at the level of Gli. Combination of NVP-LDE-225 and NVP-BEZ-235 inhibited self-renewal capacity of CSCs by suppressing the expression of pluripotency maintaining factors Nanog, Oct-4, Sox-2 and c-Myc, and transcription of Gli. NVP-LDE-225 co-operated with NVP-BEZ-235 to inhibit Lin28/Let7a/Kras axis in pancreatic CSCs. Furthermore, a superior interaction of these drugs was observed on spheroid formation by pancreatic CSCs isolated from Pankras/p53 mice. The combination of these drugs also showed superior effects on the expression of proteins involved in cell proliferation, survival and apoptosis. In addition, NVP-LDE-225 co-operated with NVP-BEZ-235 in inhibiting EMT through modulation of cadherin, vimentin and transcription factors Snail, Slug and Zeb1. In conclusion, these data suggest that the combined inhibition of PI3K/Akt/mTOR and Shh pathways may be beneficial for the treatment of pancreatic cancer.
Keywords: Gli; PI3K/AKT/mTOR; cancer stem cell; pancreatic cancer; sonic hedgehog.
Conflict of interest statement
All the authors of this article have declared “no conflict of interest”.
Figures









References
-
- Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA: a cancer journal for clinicians. 2014;64:9–29. - PubMed
-
- Adhikari AS, Agarwal N, Iwakuma T. Metastatic potential of tumor-initiating cells in solid tumors. Front Biosci. 2011;16:1927–1938. - PubMed
-
- Wang Z, Li Y, Ahmad A, Banerjee S, Azmi AS, Kong D, Sarkar FH. Pancreatic cancer: understanding and overcoming chemoresistance. Nature reviews Gastroenterology & hepatology. 2011;8:27–33. - PubMed
-
- Nusslein-Volhard C, Wieschaus E. Mutations affecting segment number and polarity in Drosophila. Nature. 1980;287:795–801. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

