Extrarenal Progenitor Cells Do Not Contribute to Renal Endothelial Repair
- PMID: 26453608
- PMCID: PMC4884107
- DOI: 10.1681/ASN.2015030321
Extrarenal Progenitor Cells Do Not Contribute to Renal Endothelial Repair
Abstract
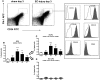
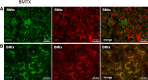
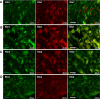
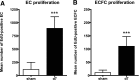
Endothelial progenitor cells (EPCs) may be relevant contributors to endothelial cell (EC) repair in various organ systems. In this study, we investigated the potential role of EPCs in renal EC repair. We analyzed the major EPC subtypes in murine kidneys, blood, and spleens after induction of selective EC injury using the concanavalin A/anti-concanavalin A model and after ischemia/reperfusion (I/R) injury as well as the potential of extrarenal cells to substitute for injured local EC. Bone marrow transplantation (BMTx), kidney transplantation, or a combination of both were performed before EC injury to allow distinction of extrarenal or BM-derived cells from intrinsic renal cells. During endothelial regeneration, cells expressing markers of endothelial colony-forming cells (ECFCs) were the most abundant EPC subtype in kidneys, but were not detected in blood or spleen. Few cells expressing markers of EC colony-forming units (EC-CFUs) were detected. In BM chimeric mice (C57BL/6 with tandem dimer Tomato-positive [tdT+] BM cells), circulating and splenic EC-CFUs were BM-derived (tdT+), whereas cells positive for ECFC markers in kidneys were not. Indeed, most BM-derived tdT+ cells in injured kidneys were inflammatory cells. Kidneys from C57BL/6 donors transplanted into tdT+ recipients with or without prior BMTx from C57BL/6 mice were negative for BM-derived or extrarenal ECFCs. Overall, extrarenal cells did not substitute for any intrinsic ECs. These results demonstrate that endothelial repair in mouse kidneys with acute endothelial lesions depends exclusively on local mechanisms.
Keywords: endothelial injury; endothelial progenitor cell; endothelial repair; glomerulonephritis; thrombotic.
Copyright © 2016 by the American Society of Nephrology.
Figures




Similar articles
-
Identification of mouse colony-forming endothelial progenitor cells for postnatal neovascularization: a novel insight highlighted by new mouse colony-forming assay.Stem Cell Res Ther. 2013 Feb 28;4(1):20. doi: 10.1186/scrt168. Stem Cell Res Ther. 2013. PMID: 23448126 Free PMC article.
-
Bone marrow monocyte lineage cells adhere on injured endothelium in a monocyte chemoattractant protein-1-dependent manner and accelerate reendothelialization as endothelial progenitor cells.Circ Res. 2003 Nov 14;93(10):980-9. doi: 10.1161/01.RES.0000099245.08637.CE. Epub 2003 Oct 2. Circ Res. 2003. PMID: 14525810
-
[Recovery of vascular niche in bone marrow by donor derived endothelial progenitor cells after allogeneic bone marrow transplantation in mice].Zhonghua Xue Ye Xue Za Zhi. 2012 Aug;33(8):623-7. Zhonghua Xue Ye Xue Za Zhi. 2012. PMID: 23134854 Chinese.
-
Bone marrow cell therapies for endothelial repair and their relevance to kidney disease.Semin Nephrol. 2012 Mar;32(2):215-23. doi: 10.1016/j.semnephrol.2012.02.008. Semin Nephrol. 2012. PMID: 22617771 Free PMC article. Review.
-
Circulating endothelial cells and circulating endothelial progenitors in kidney disease--victims, witnesses, or accomplices?Folia Biol (Praha). 2008;54(3):73-80. Folia Biol (Praha). 2008. PMID: 18647546 Review.
Cited by
-
Endothelial Colony Forming Cells (ECFCs) in murine AKI - implications for future cell-based therapies.BMC Nephrol. 2017 Feb 6;18(1):53. doi: 10.1186/s12882-017-0471-3. BMC Nephrol. 2017. PMID: 28166726 Free PMC article.
-
Interference with Gsα-Coupled Receptor Signaling in Renin-Producing Cells Leads to Renal Endothelial Damage.J Am Soc Nephrol. 2017 Dec;28(12):3479-3489. doi: 10.1681/ASN.2017020173. Epub 2017 Aug 3. J Am Soc Nephrol. 2017. PMID: 28775003 Free PMC article.
-
Peritubular Capillary Rarefaction: An Underappreciated Regulator of CKD Progression.Int J Mol Sci. 2020 Nov 4;21(21):8255. doi: 10.3390/ijms21218255. Int J Mol Sci. 2020. PMID: 33158122 Free PMC article. Review.
-
Serial intravital imaging captures dynamic and functional endothelial remodeling with single-cell resolution.JCI Insight. 2021 May 24;6(10):e123392. doi: 10.1172/jci.insight.123392. JCI Insight. 2021. PMID: 33848265 Free PMC article.
-
Role of CXCR2 in the Ac-PGP-Induced Mobilization of Circulating Angiogenic Cells and its Therapeutic Implications.Stem Cells Transl Med. 2019 Mar;8(3):236-246. doi: 10.1002/sctm.18-0035. Epub 2018 Nov 26. Stem Cells Transl Med. 2019. PMID: 30474937 Free PMC article.
References
-
- Legendre CM, Licht C, Muus P, Greenbaum LA, Babu S, Bedrosian C, Bingham C, Cohen DJ, Delmas Y, Douglas K, Eitner F, Feldkamp T, Fouque D, Furman RR, Gaber O, Herthelius M, Hourmant M, Karpman D, Lebranchu Y, Mariat C, Menne J, Moulin B, Nürnberger J, Ogawa M, Remuzzi G, Richard T, Sberro-Soussan R, Severino B, Sheerin NS, Trivelli A, Zimmerhackl LB, Goodship T, Loirat C: Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N Engl J Med 368: 2169–2181, 2013 - PubMed
-
- Chapman K, Seldon M, Richards R: Thrombotic microangiopathies, thrombotic thrombocytopenic purpura, and ADAMTS-13. Semin Thromb Hemost 38: 47–54, 2012 - PubMed
-
- Dong JF, Moake JL, Nolasco L, Bernardo A, Arceneaux W, Shrimpton CN, Schade AJ, McIntire LV, Fujikawa K, López JA: ADAMTS-13 rapidly cleaves newly secreted ultralarge von Willebrand factor multimers on the endothelial surface under flowing conditions. Blood 100: 4033–4039, 2002 - PubMed
-
- Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, Witzenbichler B, Schatteman G, Isner JM: Isolation of putative progenitor endothelial cells for angiogenesis. Science 275: 964–967, 1997 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

