4-Hydroxynonenal activates Src through a non-canonical pathway that involves EGFR/PTP1B
- PMID: 26453921
- PMCID: PMC4684732
- DOI: 10.1016/j.freeradbiomed.2015.08.025
4-Hydroxynonenal activates Src through a non-canonical pathway that involves EGFR/PTP1B
Abstract
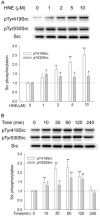
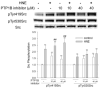
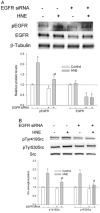
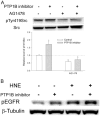
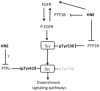
Src, a non-receptor protein tyrosine kinase involved in many biological processes, can be activated through both redox-dependent and independent mechanisms. 4-Hydroxy-2-nonenal (HNE) is a lipid peroxidation product that is increased in pathophysiological conditions associated with Src activation. This study examined how HNE activates human c-Src. In the canonical pathway Src activation is initiated by dephosphorylation of pTyr530 followed by conformational change that causes Src auto-phosphorylation at Tyr419 and its activation. HNE increased Src activation in both dose- and time-dependent manner, while it also increased Src phosphorylation at Tyr530 (pTyr530 Src), suggesting that HNE activated Src via a non-canonical mechanism. Protein tyrosine phosphatase 1B inhibitor (539741), at concentrations that increased basal pTyr530 Src, also increased basal Src activity and significantly reduced HNE-mediated Src activation. The EGFR inhibitor, AG1478, and EGFR silencing, abrogated HNE-mediated EGFR activation and inhibited basal and HNE-induced Src activity. In addition, AG1478 also eliminated the increase of basal Src activation by a PTP1B inhibitor. Taken together these data suggest that HNE can activate Src partly through a non-canonical pathway involving activation of EGFR and inhibition of PTP1B.
Keywords: EGFR; Src kinase; lipid peroxidation; protein tyrosine phosphatase; redox signaling; signal transduction.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
TGFβ1 rapidly activates Src through a non-canonical redox signaling mechanism.Arch Biochem Biophys. 2015 Feb 15;568:1-7. doi: 10.1016/j.abb.2015.01.001. Epub 2015 Jan 10. Arch Biochem Biophys. 2015. PMID: 25585026 Free PMC article.
-
Src Tyrosine Kinase Activation by 4-Hydroxynonenal Upregulates p38, ERK/AP-1 Signaling and COX-2 Expression in YPEN-1 Cells.PLoS One. 2015 Oct 14;10(10):e0129244. doi: 10.1371/journal.pone.0129244. eCollection 2015. PLoS One. 2015. PMID: 26466383 Free PMC article.
-
Radiation-induced lipid peroxidation activates src kinase and triggers nuclear EGFR transport.Radiother Oncol. 2009 Sep;92(3):379-82. doi: 10.1016/j.radonc.2009.06.003. Epub 2009 Jun 25. Radiother Oncol. 2009. PMID: 19560222
-
4-hydroxynonenal triggers multistep signal transduction cascades for suppression of cellular functions.Mol Aspects Med. 2003 Aug-Oct;24(4-5):231-8. doi: 10.1016/s0098-2997(03)00018-9. Mol Aspects Med. 2003. PMID: 12893001 Review.
-
Oxidized LDL and 4-hydroxynonenal modulate tyrosine kinase receptor activity.Mol Aspects Med. 2003 Aug-Oct;24(4-5):251-61. doi: 10.1016/s0098-2997(03)00020-7. Mol Aspects Med. 2003. PMID: 12893003 Review.
Cited by
-
Activation of proinflammatory signaling by 4-hydroxynonenal-Src adducts in aged kidneys.Oncotarget. 2016 Aug 9;7(32):50864-50874. doi: 10.18632/oncotarget.10854. Oncotarget. 2016. PMID: 27472463 Free PMC article.
-
Reactive Carbonyl Species and Protein Lipoxidation in Atherogenesis.Antioxidants (Basel). 2024 Feb 14;13(2):232. doi: 10.3390/antiox13020232. Antioxidants (Basel). 2024. PMID: 38397830 Free PMC article. Review.
-
Direct 1O2 optical excitation: A tool for redox biology.Redox Biol. 2017 Oct;13:39-59. doi: 10.1016/j.redox.2017.05.011. Epub 2017 May 25. Redox Biol. 2017. PMID: 28570948 Free PMC article. Review.
-
4-Hydroxynonenal dependent alteration of TRPV1-mediated coronary microvascular signaling.Free Radic Biol Med. 2016 Dec;101:10-19. doi: 10.1016/j.freeradbiomed.2016.09.021. Epub 2016 Sep 25. Free Radic Biol Med. 2016. PMID: 27682362 Free PMC article.
-
Identification of Novel Src Inhibitors: Pharmacophore-Based Virtual Screening, Molecular Docking and Molecular Dynamics Simulations.Molecules. 2020 Sep 8;25(18):4094. doi: 10.3390/molecules25184094. Molecules. 2020. PMID: 32911607 Free PMC article.
References
-
- Parsons SJ, Parsons JT. Src family kinases, key regulators of signal transduction. Oncogene. 2004;23:7906–7909. - PubMed
-
- Guarino M. Src signaling in cancer invasion. Journal of cellular physiology. 2010;223:14–26. - PubMed
-
- Lutz MP, Esser IB, Flossmann-Kast BB, Vogelmann R, Luhrs H, Friess H, Buchler MW, Adler G. Overexpression and activation of the tyrosine kinase Src in human pancreatic carcinoma. Biochemical and biophysical research communications. 1998;243:503–508. - PubMed
-
- Summy JM, Gallick GE. Src family kinases in tumor progression and metastasis. Cancer metastasis reviews. 2003;22:337–358. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

