Mature adipocytes in bone marrow protect myeloma cells against chemotherapy through autophagy activation
- PMID: 26455377
- PMCID: PMC4741456
- DOI: 10.18632/oncotarget.6020
Mature adipocytes in bone marrow protect myeloma cells against chemotherapy through autophagy activation
Abstract
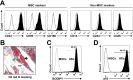
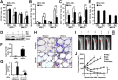
A major problem in patients with multiple myeloma is chemotherapy resistance, which develops in myeloma cells upon interaction with bone marrow stromal cells. However, few studies have determined the role of bone marrow adipocytes, a major component of stromal cells in the bone marrow, in myeloma chemotherapy resistance. We reveal that mature human adipocytes activate autophagy and upregulate the expression of autophagic proteins, thereby suppressing chemotherapy-induced caspase cleavage and apoptosis in myeloma cells. We found that adipocytes secreted known and novel adipokines, such as leptin and adipsin. The addition of these adipokines enhanced the expression of autophagic proteins and reduced apoptosis in myeloma cells. In vivo studies further demonstrated the importance of bone marrow-derived adipocytes in the reduced response of myeloma cells to chemotherapy. Our findings suggest that adipocytes, adipocyte-secreted adipokines, and adipocyte-activated autophagy are novel targets for combatting chemotherapy resistance and enhancing treatment efficacy in myeloma patients.
Keywords: adipocytes; apoptosis; autophagy; chemotherapy resistance; multiple myeloma.
Conflict of interest statement
The authors have no competing financial or intellectual interests.
Figures





References
-
- Kyle RA, Rajkumar SV. Multiple myeloma. N Engl J Med. 2004;351:1860–1873. - PubMed
-
- Dispenzieri A, Kyle RA. Multiple myeloma: clinical features and indications for therapy. Best Pract Res Clin Haematol. 2005;18:553–568. - PubMed
-
- Rajkumar SV. Novel approaches to the management of myeloma. Oncology (Williston Park) 2005;19:621–625. - PubMed
-
- Shaughnessy JD., Jr and Barlogie B. Using genomics to identify high-risk myeloma after autologous stem cell transplantation. Biol Blood Marrow Transplant. 2006;12:77–80. - PubMed
-
- Durie BG, Waxman AD, D'Agnolo A, Williams CM. Whole-body (18) F-FDG PET identifies high-risk myeloma. J Nucl Med. 2002;43:1457–1463. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

