Macrophage-derived reactive oxygen species protects against autoimmune priming with a defined polymeric adjuvant
- PMID: 26455429
- PMCID: PMC4693874
- DOI: 10.1111/imm.12546
Macrophage-derived reactive oxygen species protects against autoimmune priming with a defined polymeric adjuvant
Abstract
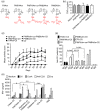
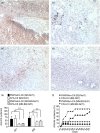
Understanding the nature of adjuvants and the immune priming events in autoimmune diseases, such as rheumatoid arthritis, is a key challenge to identify their aetiology. Adjuvants are, however, complex structures with inflammatory and immune priming properties. Synthetic polymers provide a possibility to separate these functions and allow studies of the priming mechanisms in vivo. A well-balanced polymer, poly-N-isopropyl acrylamide (PNiPAAm) mixed with collagen type II (CII) induced relatively stronger autoimmunity and arthritis compared with more hydrophilic (polyacrylamide) or hydrophobic (poly-N-isopropylacrylamide-co-poly-N-tertbutylacrylamide and poly-N-tertbutylacrylamide) polymers. Clearly, all the synthesized polymers except the more hydrophobic poly-N-tertbutylacrylamide induced arthritis, especially in Ncf1-deficient mice, which are deficient in reactive oxygen species (ROS) production. We identified macrophages as the major infiltrating cells present at PNiPAAm-CII injection sites and demonstrate that ROS produced by the macrophages attenuated the immune response and the development of arthritis. Our results reveal that thermo-responsive polymers with high immune priming capacity could trigger an autoimmune response to CII and the subsequent arthritis development, in particular in the absence of NOX2 derived ROS. Importantly, ROS from macrophages protected against the autoimmune priming, demonstrating a critical regulatory role of macrophages in immune priming events.
Keywords: adjuvant; arthritis; collagen type II; macrophages; poly-N-isopropylacrylamide.
© 2015 John Wiley & Sons Ltd.
Figures

References
-
- Shakya AK, Holmdahl R, Nandakumar KS, Kumar A. Characterization of chemically defined poly‐N‐isopropylacrylamide based copolymeric adjuvants. Vaccine 2013; 31:3519–27. - PubMed
-
- Shakya AK, Nandakumar KS. Synthetic polymer as an adjuvant in collagen‐induced arthritis. Curr Protoc Mouse Biol 2014; 4:11–24. - PubMed
-
- Shakya AK, Kumar A, Klaczkowska D, Hultqvist M, Hagenow K, Holmdahl R et al Collagen type II and a thermo‐responsive polymer of N‐isopropylacrylamide induce arthritis independent of Toll‐like receptors: a strong influence by major histocompatibility complex class II and Ncf1 genes. Am J Pathol 2011; 179:2490–500. - PMC - PubMed
-
- Shakya AK, Nandkumar KS. Polymers as immunological adjuvants: an update on recent developments. J Biosci Biotechnol 2012; 1:199–210.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

