The broad-spectrum antiviral favipiravir protects guinea pigs from lethal Lassa virus infection post-disease onset
- PMID: 26456301
- PMCID: PMC4600983
- DOI: 10.1038/srep14775
The broad-spectrum antiviral favipiravir protects guinea pigs from lethal Lassa virus infection post-disease onset
Abstract
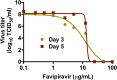
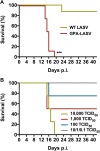
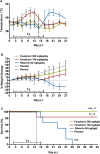
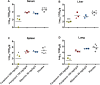
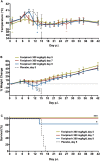
With up to 500,000 infections annually, Lassa virus (LASV), the cause of Lassa fever, is one of the most prevalent etiological agents of viral hemorrhagic fever (VHF) in humans. LASV is endemic in several West African countries with sporadic cases and prolonged outbreaks observed most commonly in Sierra Leone, Liberia, Guinea and Nigeria. Additionally several cases of Lassa fever have been imported into North America, Europe and Asia making LASV a global threat to public health. Despite this, currently no approved therapeutic or vaccine exists to treat or prevent LASV infections. Here, using a passaged strain of LASV that is uniformly lethal in Hartley guinea pigs, we demonstrate that favipiravir, a broad-spectrum antiviral agent and leading treatment option for influenza, has potent activity against LASV infection. In this model, once daily treatment with favipiravir significantly reduced viral titers in tissue samples and reduced mortality rates when compared with animals receiving vehicle-only or ribavirin, the current standard of care for Lassa fever. Favipiravir remained highly effective against lethal LASV infection when treatments were initiated nine days post-infection, a time when animals were demonstrating advanced signs of disease. These results support the further preclinical evaluation of favipiravir for Lassa fever and other VHFs.
Conflict of interest statement
Y.F. and T.K. are employees of the Toyama Chemical Co., Ltd., the manufacturer of favipiravir. All other authors declare that no competing interests exist.
Figures

Similar articles
-
Lethal Infection of Lassa Virus Isolated from a Human Clinical Sample in Outbred Guinea Pigs without Adaptation.mSphere. 2019 Sep 25;4(5):e00428-19. doi: 10.1128/mSphere.00428-19. mSphere. 2019. PMID: 31554720 Free PMC article.
-
Lassa viral dynamics in non-human primates treated with favipiravir or ribavirin.PLoS Comput Biol. 2021 Jan 7;17(1):e1008535. doi: 10.1371/journal.pcbi.1008535. eCollection 2021 Jan. PLoS Comput Biol. 2021. PMID: 33411731 Free PMC article.
-
A recombinant vesicular stomatitis virus-based Lassa fever vaccine protects guinea pigs and macaques against challenge with geographically and genetically distinct Lassa viruses.PLoS Negl Trop Dis. 2015 Apr 17;9(4):e0003736. doi: 10.1371/journal.pntd.0003736. eCollection 2015 Apr. PLoS Negl Trop Dis. 2015. PMID: 25884628 Free PMC article.
-
Current research for a vaccine against Lassa hemorrhagic fever virus.Drug Des Devel Ther. 2018 Aug 14;12:2519-2527. doi: 10.2147/DDDT.S147276. eCollection 2018. Drug Des Devel Ther. 2018. PMID: 30147299 Free PMC article. Review.
-
New opportunities for field research on the pathogenesis and treatment of Lassa fever.Antiviral Res. 2008 Apr;78(1):103-15. doi: 10.1016/j.antiviral.2007.11.003. Epub 2007 Dec 17. Antiviral Res. 2008. PMID: 18241935 Review.
Cited by
-
Severe mammarenaviral disease in guinea pigs effectively treated by an orally bioavailable fusion inhibitor, alone or in combination with favipiravir.Antiviral Res. 2022 Dec;208:105444. doi: 10.1016/j.antiviral.2022.105444. Epub 2022 Oct 13. Antiviral Res. 2022. PMID: 36243175 Free PMC article.
-
Combating Lassa Fever in West African Sub-Region: Progress, Challenges, and Future Perspectives.Viruses. 2023 Jan 3;15(1):146. doi: 10.3390/v15010146. Viruses. 2023. PMID: 36680186 Free PMC article. Review.
-
Recombinant Lassa Virus Expressing Green Fluorescent Protein as a Tool for High-Throughput Drug Screens and Neutralizing Antibody Assays.Viruses. 2018 Nov 20;10(11):655. doi: 10.3390/v10110655. Viruses. 2018. PMID: 30463334 Free PMC article.
-
Favipiravir (T-705) but not ribavirin is effective against two distinct strains of Crimean-Congo hemorrhagic fever virus in mice.Antiviral Res. 2018 Sep;157:18-26. doi: 10.1016/j.antiviral.2018.06.013. Epub 2018 Jun 21. Antiviral Res. 2018. PMID: 29936152 Free PMC article.
-
Lassa Virus Treatment Options.Microorganisms. 2021 Apr 7;9(4):772. doi: 10.3390/microorganisms9040772. Microorganisms. 2021. PMID: 33917071 Free PMC article. Review.
References
-
- McCormick J. B. & Fisher-Hoch S. P. Lassa fever. Current topics in microbiology and immunology 262, 75–109 (2002). - PubMed
-
- McCormick J. B. et al. A case-control study of the clinical diagnosis and course of Lassa fever. The Journal of infectious diseases 155, 445–455 (1987). - PubMed
-
- McCormick J. B. Epidemiology and control of Lassa fever. Current topics in microbiology and immunology 134, 69–78 (1987). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

