β-Catenin Upregulates the Constitutive and Virus-Induced Transcriptional Capacity of the Interferon Beta Promoter through T-Cell Factor Binding Sites
- PMID: 26459757
- PMCID: PMC4702592
- DOI: 10.1128/MCB.00641-15
β-Catenin Upregulates the Constitutive and Virus-Induced Transcriptional Capacity of the Interferon Beta Promoter through T-Cell Factor Binding Sites
Abstract
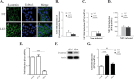
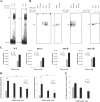
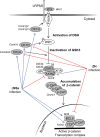
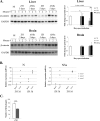
Rapid upregulation of interferon beta (IFN-β) expression following virus infection is essential to set up an efficient innate antiviral response. Biological roles related to the antiviral and immune response have also been associated with the constitutive production of IFN-β in naive cells. However, the mechanisms capable of modulating constitutive IFN-β expression in the absence of infection remain largely unknown. In this work, we demonstrate that inhibition of the kinase glycogen synthase kinase 3 (GSK-3) leads to the upregulation of the constitutive level of IFN-β expression in noninfected cells, provided that GSK-3 inhibition is correlated with the binding of β-catenin to the IFN-β promoter. Under these conditions, IFN-β expression occurred through the T-cell factor (TCF) binding sites present on the IFN-β promoter independently of interferon regulatory factor 3 (IRF3). Enhancement of the constitutive level of IFN-β per se was able to confer an efficient antiviral state to naive cells and acted in synergy with virus infection to stimulate virus-induced IFN-β expression. Further emphasizing the role of β-catenin in the innate antiviral response, we show here that highly pathogenic Rift Valley fever virus (RVFV) targets the Wnt/β-catenin pathway and the formation of active TCF/β-catenin complexes at the transcriptional and protein level in RVFV-infected cells and mice.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures




Similar articles
-
Regulation of human nitric oxide synthase 2 expression by Wnt beta-catenin signaling.Cancer Res. 2006 Jul 15;66(14):7024-31. doi: 10.1158/0008-5472.CAN-05-4110. Cancer Res. 2006. PMID: 16849547
-
Fine-Tuning of the RIG-I-Like Receptor/Interferon Regulatory Factor 3-Dependent Antiviral Innate Immune Response by the Glycogen Synthase Kinase 3/β-Catenin Pathway.Mol Cell Biol. 2015 Sep 1;35(17):3029-43. doi: 10.1128/MCB.00344-15. Epub 2015 Jun 22. Mol Cell Biol. 2015. PMID: 26100021 Free PMC article.
-
Roles of GSK-3 and β-Catenin in Antiviral Innate Immune Sensing of Nucleic Acids.Cells. 2020 Apr 7;9(4):897. doi: 10.3390/cells9040897. Cells. 2020. PMID: 32272583 Free PMC article. Review.
-
A Tcf/Lef element within the enhancer region of the human NANOG gene plays a role in promoter activation.Biochem Biophys Res Commun. 2011 Jul 8;410(3):637-42. doi: 10.1016/j.bbrc.2011.06.044. Epub 2011 Jun 13. Biochem Biophys Res Commun. 2011. PMID: 21689639
-
Bone morphogenetic protein-2 enhances Wnt/beta-catenin signaling-induced osteoprotegerin expression.Genes Cells. 2009 Feb;14(2):141-53. doi: 10.1111/j.1365-2443.2008.01258.x. Epub 2008 Jan 6. Genes Cells. 2009. PMID: 19170762
Cited by
-
A Genome-Wide RNA Interference Screen Identifies a Role for Wnt/β-Catenin Signaling during Rift Valley Fever Virus Infection.J Virol. 2016 Jul 27;90(16):7084-7097. doi: 10.1128/JVI.00543-16. Print 2016 Aug 15. J Virol. 2016. PMID: 27226375 Free PMC article.
-
LINC01002 functions as a ceRNA to regulate FRMD8 by sponging miR-4324 for the development of COVID-19.Virol J. 2024 May 11;21(1):109. doi: 10.1186/s12985-024-02382-2. Virol J. 2024. PMID: 38734674 Free PMC article.
-
Phosphorylation of Shrimp Tcf by a Viral Protein Kinase WSV083 Suppresses Its Antiviral Effect.Front Immunol. 2021 Aug 2;12:698697. doi: 10.3389/fimmu.2021.698697. eCollection 2021. Front Immunol. 2021. PMID: 34408747 Free PMC article.
-
Differential regulation of the Wnt/β-catenin pathway by hepatitis C virus recombinants expressing core from various genotypes.Sci Rep. 2018 Jul 25;8(1):11185. doi: 10.1038/s41598-018-29078-2. Sci Rep. 2018. PMID: 30046100 Free PMC article.
-
Regulation of Wnt/β-catenin signaling by herpesviruses.World J Virol. 2016 Nov 12;5(4):144-154. doi: 10.5501/wjv.v5.i4.144. World J Virol. 2016. PMID: 27878101 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
