Unconstrained muscle-tendon workloops indicate resonance tuning as a mechanism for elastic limb behavior during terrestrial locomotion
- PMID: 26460038
- PMCID: PMC4629365
- DOI: 10.1073/pnas.1500702112
Unconstrained muscle-tendon workloops indicate resonance tuning as a mechanism for elastic limb behavior during terrestrial locomotion
Abstract
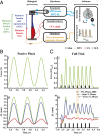
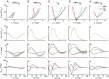
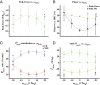
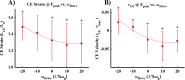
In terrestrial locomotion, there is a missing link between observed spring-like limb mechanics and the physiological systems driving their emergence. Previous modeling and experimental studies of bouncing gait (e.g., walking, running, hopping) identified muscle-tendon interactions that cycle large amounts of energy in series tendon as a source of elastic limb behavior. The neural, biomechanical, and environmental origins of these tuned mechanics, however, have remained elusive. To examine the dynamic interplay between these factors, we developed an experimental platform comprised of a feedback-controlled servo-motor coupled to a biological muscle-tendon. Our novel motor controller mimicked in vivo inertial/gravitational loading experienced by muscles during terrestrial locomotion, and rhythmic patterns of muscle activation were applied via stimulation of intact nerve. This approach was based on classical workloop studies, but avoided predetermined patterns of muscle strain and activation-constraints not imposed during real-world locomotion. Our unconstrained approach to position control allowed observation of emergent muscle-tendon mechanics resulting from dynamic interaction of neural control, active muscle, and system material/inertial properties. This study demonstrated that, despite the complex nonlinear nature of musculotendon systems, cyclic muscle contractions at the passive natural frequency of the underlying biomechanical system yielded maximal forces and fractions of mechanical work recovered from previously stored elastic energy in series-compliant tissues. By matching movement frequency to the natural frequency of the passive biomechanical system (i.e., resonance tuning), muscle-tendon interactions resulting in spring-like behavior emerged naturally, without closed-loop neural control. This conceptual framework may explain the basis for elastic limb behavior during terrestrial locomotion.
Keywords: elastic limb behavior; muscle-tendon mechanics; neural control; resonance; terrestrial locomotion.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Timing matters: tuning the mechanics of a muscle-tendon unit by adjusting stimulation phase during cyclic contractions.J Exp Biol. 2015 Oct;218(Pt 19):3150-9. doi: 10.1242/jeb.121673. Epub 2015 Jul 31. J Exp Biol. 2015. PMID: 26232413 Free PMC article.
-
A benchtop biorobotic platform for in vitro observation of muscle-tendon dynamics with parallel mechanical assistance from an elastic exoskeleton.J Biomech. 2017 May 24;57:8-17. doi: 10.1016/j.jbiomech.2017.03.009. Epub 2017 Mar 21. J Biomech. 2017. PMID: 28431748
-
Exploiting elasticity: Modeling the influence of neural control on mechanics and energetics of ankle muscle-tendons during human hopping.J Theor Biol. 2014 Jul 21;353:121-32. doi: 10.1016/j.jtbi.2014.03.010. Epub 2014 Mar 16. J Theor Biol. 2014. PMID: 24641822
-
Patterns of mechanical energy change in tetrapod gait: pendula, springs and work.J Exp Zool A Comp Exp Biol. 2006 Nov 1;305(11):899-911. doi: 10.1002/jez.a.334. J Exp Zool A Comp Exp Biol. 2006. PMID: 17029267 Review.
-
Muscle-tendon stresses and elastic energy storage during locomotion in the horse.Comp Biochem Physiol B Biochem Mol Biol. 1998 May;120(1):73-87. doi: 10.1016/s0305-0491(98)00024-8. Comp Biochem Physiol B Biochem Mol Biol. 1998. PMID: 9787779 Review.
Cited by
-
Some Challenges of Playing with Power: Does Complex Energy Flow Constrain Neuromuscular Performance?Integr Comp Biol. 2019 Dec 1;59(6):1619-1628. doi: 10.1093/icb/icz108. Integr Comp Biol. 2019. PMID: 31241134 Free PMC article.
-
Tuning of feedforward control enables stable muscle force-length dynamics after loss of autogenic proprioceptive feedback.Elife. 2020 Jun 23;9:e53908. doi: 10.7554/eLife.53908. Elife. 2020. PMID: 32573432 Free PMC article.
-
Humans falling in holes: adaptations in lower-limb joint mechanics in response to a rapid change in substrate height during human hopping.J R Soc Interface. 2019 Oct 31;16(159):20190292. doi: 10.1098/rsif.2019.0292. Epub 2019 Oct 2. J R Soc Interface. 2019. PMID: 31575349 Free PMC article. Clinical Trial.
-
A Conceptual Exploration of Hamstring Muscle-Tendon Functioning during the Late-Swing Phase of Sprinting: The Importance of Evidence-Based Hamstring Training Frameworks.Sports Med. 2023 Dec;53(12):2321-2346. doi: 10.1007/s40279-023-01904-2. Epub 2023 Sep 5. Sports Med. 2023. PMID: 37668895 Free PMC article. Review.
-
Linking in vivo muscle dynamics to force-length and force-velocity properties reveals that guinea fowl lateral gastrocnemius operates at shorter than optimal lengths.J Exp Biol. 2024 Aug 1;227(15):jeb246879. doi: 10.1242/jeb.246879. Epub 2024 Aug 1. J Exp Biol. 2024. PMID: 38873800 Free PMC article.
References
-
- Farley CT, Glasheen J, McMahon TA. Running springs: Speed and animal size. J Exp Biol. 1993;185:71–86. - PubMed
-
- Farley CT, González O. Leg stiffness and stride frequency in human running. J Biomech. 1996;29(2):181–186. - PubMed
-
- Blickhan R. The spring-mass model for running and hopping. J Biomech. 1989;22(11-12):1217–1227. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

