Discovery of m(7)G-cap in eukaryotic mRNAs
- PMID: 26460318
- PMCID: PMC4729855
- DOI: 10.2183/pjab.91.394
Discovery of m(7)G-cap in eukaryotic mRNAs
Abstract
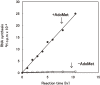
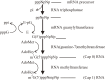
Terminal structure analysis of an insect cytoplasmic polyhedrosis virus (CPV) genome RNA in the early 1970s at the National Institute of Genetics in Japan yielded a 2'-O-methylated nucleotide in the 5' end of double-stranded RNA genome. This finding prompted me to add S-adenosyl-L-methionine, a natural methylation donor, to the in vitro transcription reaction of viruses that contain RNA polymerase. This effort resulted in unprecedented mRNA synthesis that generates a unique blocked and methylated 5' terminal structure (referred later to as "cap" or "m(7)G-cap") in the transcription of silkworm CPV and human reovirus and vaccinia viruses that contain RNA polymerase in virus particles. Initial studies with viruses paved the way to discover the 5'-cap m(7)GpppNm structure present generally in cellular mRNAs of eukaryotes. I participated in those studies and was able to explain the pathway of cap synthesis and the significance of the 5' cap (and capping) in gene expression processes, including transcription and protein synthesis. In this review article I concentrate on the description of these initial studies that eventually led us to a new paradigm of mRNA capping.
Figures




References
-
- Furuichi, Y. (2014) Caps on eukaryotic mRNAs. In eLS. John Wiley and Sons, Ltd., Chichester. doi: 10.1002/9780470015902.a0000891.pub3.
-
- Weaver, R.F. (2008) Messenger RNA and processing II: Capping and Polyadenylation. In Molecular Biology. Fourth Edition, McGraw-Hill Companies, Inc., New York, Chapter 15.
-
- Furuichi Y., Shatkin A.J. (1989) Characterization of cap structure. Methods Enzymol. 180, 164–177. - PubMed
-
- Furuichi Y., LaFiandra A., Shatkin A.J. (1977) 5′-Terminal structure and mRNA stability. Nature 266, 235–239. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

