Differential expression of key regulators of Toll-like receptors in ulcerative colitis and Crohn's disease: a role for Tollip and peroxisome proliferator-activated receptor gamma?
- PMID: 26462859
- PMCID: PMC4750602
- DOI: 10.1111/cei.12732
Differential expression of key regulators of Toll-like receptors in ulcerative colitis and Crohn's disease: a role for Tollip and peroxisome proliferator-activated receptor gamma?
Abstract
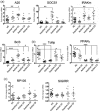
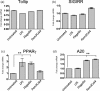
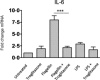
The innate immune system is currently seen as the probable initiator of events which culminate in the development of inflammatory bowel disease (IBD) with Toll-like receptors (TLRs) known to be involved in this disease process. Many regulators of TLRs have been described, and dysregulation of these may also be important in the pathogenesis of IBD. The aim of this study was to perform a co-ordinated analysis of the expression levels of both key intestinal TLRs and their inhibitory proteins in the same IBD cohorts, both ulcerative colitis (UC) and Crohn's disease (CD), in order to evaluate the potential roles of these proteins in the pathogenesis of IBD. Of the six TLRs (TLRs 1, 2, 4, 5, 6 and 9) examined, only TLR-4 was increased significantly in IBD, specifically in active UC. In contrast, differential alterations in expression of TLR inhibitory proteins were observed. A20 and suppressor of cytokine signalling 1 (SOCS1) were increased only in active UC while interleukin-1 receptor-associated kinase 1 (IRAK-m) and B cell lymphoma 3 protein (Bcl-3) were increased in both active UC and CD. In contrast, expression of both peroxisome proliferator-activated receptor gamma (PPARγ) and Toll interacting protein (Tollip) was decreased in both active and inactive UC and CD and at both mRNA and protein levels. In addition, expression of both PPARγ and A20 expression was increased by stimulation of a colonic epithelial cell line Caco-2 with both TLR ligands and commensal bacterial strains. These data suggest that IBD may be associated with distinctive changes in TLR-4 and TLR inhibitory proteins, implying that alterations in these may contribute to the pathogenesis of IBD.
Keywords: PPARγ; Toll-like receptor; Tollip; inflammatory bowel disease; regulation.
© 2015 British Society for Immunology.
Figures



Similar articles
-
Peroxisome proliferator-activated receptor γ 2 mutation may cause a subset of ulcerative colitis.Pediatr Int. 2010 Oct;52(5):729-34. doi: 10.1111/j.1442-200X.2010.03195.x. Pediatr Int. 2010. PMID: 20591056
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
-
Expression and function of toll-like receptor 8 and Tollip in colonic epithelial cells from patients with inflammatory bowel disease.Scand J Gastroenterol. 2009;44(2):195-204. doi: 10.1080/00365520802495529. Scand J Gastroenterol. 2009. PMID: 18985539
-
Impaired expression of peroxisome proliferator-activated receptor gamma in ulcerative colitis.Gastroenterology. 2003 May;124(5):1265-76. doi: 10.1016/s0016-5085(03)00271-3. Gastroenterology. 2003. PMID: 12730867
-
TLRs in the Gut. IV. Negative regulation of Toll-like receptors and intestinal homeostasis: addition by subtraction.Am J Physiol Gastrointest Liver Physiol. 2007 Jun;292(6):G1469-73. doi: 10.1152/ajpgi.00531.2006. Am J Physiol Gastrointest Liver Physiol. 2007. PMID: 17554134 Review.
Cited by
-
TLR4 signaling in the development of colitis-associated cancer and its possible interplay with microRNA-155.Cell Commun Signal. 2021 Sep 3;19(1):90. doi: 10.1186/s12964-021-00771-6. Cell Commun Signal. 2021. PMID: 34479599 Free PMC article. Review.
-
The Versatile Role of Peroxisome Proliferator-Activated Receptors in Immune-Mediated Intestinal Diseases.Cells. 2024 Oct 12;13(20):1688. doi: 10.3390/cells13201688. Cells. 2024. PMID: 39451206 Free PMC article. Review.
-
The Contribution of Genetic and Epigenetic Factors: An Emerging Concept in the Assessment and Prognosis of Inflammatory Bowel Diseases.Int J Mol Sci. 2024 Aug 1;25(15):8420. doi: 10.3390/ijms25158420. Int J Mol Sci. 2024. PMID: 39125988 Free PMC article. Review.
-
Toll-Interacting Protein in Resolving and Non-Resolving Inflammation.Front Immunol. 2017 May 5;8:511. doi: 10.3389/fimmu.2017.00511. eCollection 2017. Front Immunol. 2017. PMID: 28529512 Free PMC article. Review.
-
TLR2 and TLR4 in Parkinson's disease pathogenesis: the environment takes a toll on the gut.Transl Neurodegener. 2021 Nov 17;10(1):47. doi: 10.1186/s40035-021-00271-0. Transl Neurodegener. 2021. PMID: 34814947 Free PMC article. Review.
References
-
- Walsh D, McCarthy J, O'Driscoll C, Melgar S. Pattern recognition receptors–molecular orchestrators of inflammation in inflammatory bowel disease. Cytokine Growth Factor Rev 2013; 24:91–104. - PubMed
-
- Shibolet O, Podolsky DK. TLRs in the gut. IV. Negative regulation of Toll‐like receptors and intestinal homeostasis: addition by subtraction. Am J Physiol Gastrointest Liver Physiol 2007; 292:G1469–73. - PubMed
-
- Regan T, Nally K, Carmody R et al Identification of TLR10 as a key mediator of the inflammatory response to Listeria monocytogenes in intestinal epithelial cells and macrophages. J Immunol 2013; 191:6084–92. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

