Significant neuronal soma volume deficit in the limbic system in subjects with 15q11.2-q13 duplications
- PMID: 26463344
- PMCID: PMC4603300
- DOI: 10.1186/s40478-015-0241-z
Significant neuronal soma volume deficit in the limbic system in subjects with 15q11.2-q13 duplications
Abstract
Introduction: Autism is diagnosed in numerous genetic and genomic developmental disorders associated with an overlap in high-risk genes and loci that underlie intellectual disability (ID) and epilepsy. The aim of this stereological study of neuronal soma volume in 25 brain structures and their subdivisions in eight individuals 9 to 26 years of age who were diagnosed with chromosome 15q11.2-13.1 duplication syndrome [dup(15)], autism, ID and epilepsy; eight age-matched subjects diagnosed with autism of unknown etiology (idiopathic autism) and seven control individuals was to establish whether defects of neuronal soma growth are a common denominator of developmental pathology in idiopathic and syndromic autism and how genetic modifications alter the trajectory of neuronal soma growth in dup(15) autism.
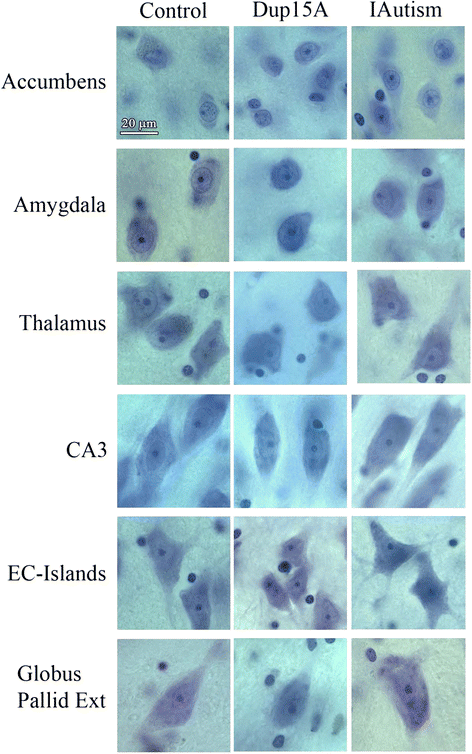
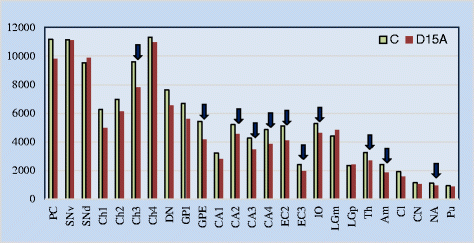
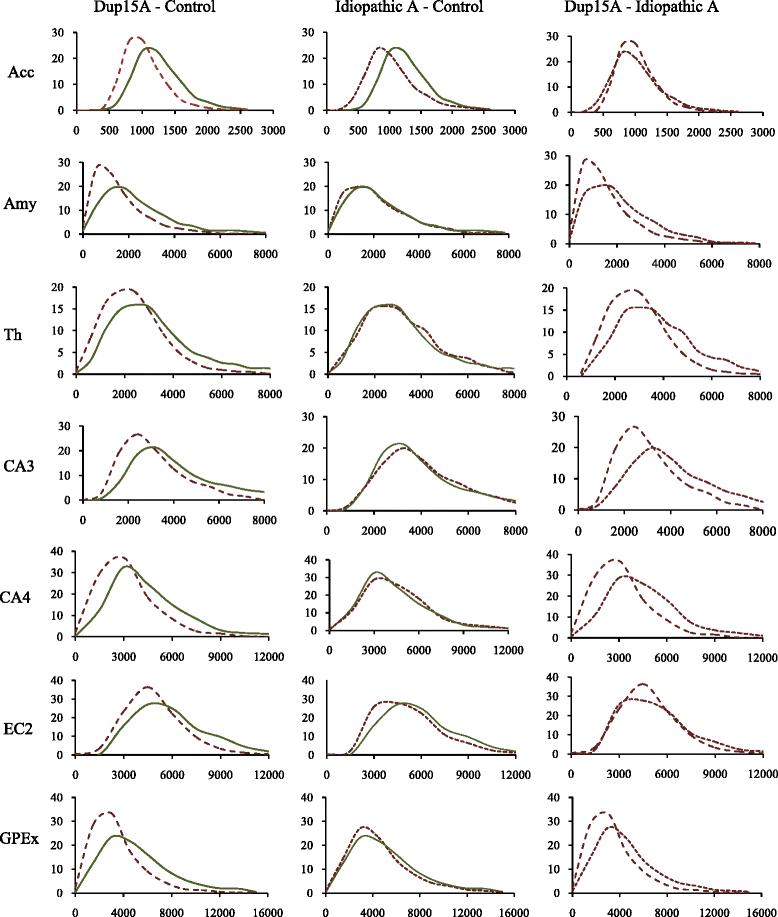
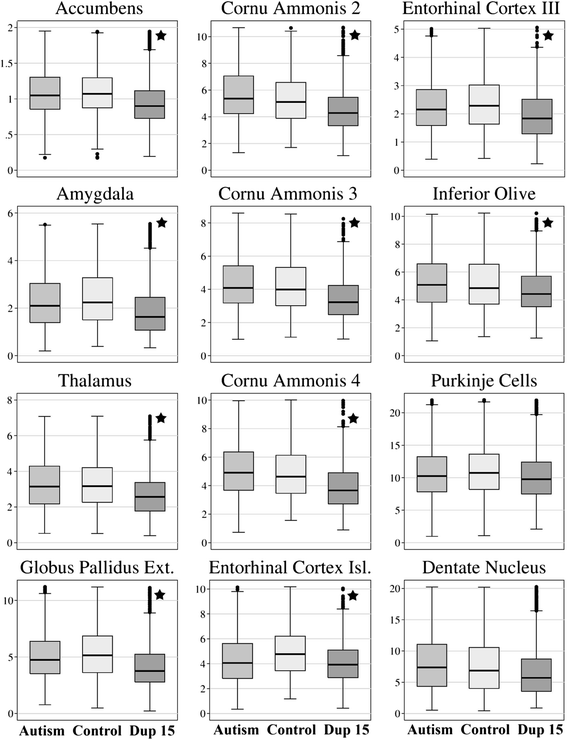
Results: Application of the Nucleator software to estimate neuronal size revealed significant neuronal soma volume deficits in 11 of 25 structures and their subregions (44 %) in subjects diagnosed with dup(15) autism, including consistent neuronal soma volume deficits in the limbic system (sectors CA2, 3 and 4 in Ammon's horn, the second and third layers of the entorhinal cortex and in the amygdala), as well as in the thalamus, nucleus accumbens, external globus pallidus, and Ch3 nucleus in the magnocellular basal complex, and in the inferior olive in the brainstem. The second feature distinguishing dup(15) autism was persistent neuronal soma deficits in adolescents and young adults, whereas in idiopathic autism, neuronal volume deficit is most prominent in 4- to 8-year-old children but affects only a few brain regions in older subjects.
Conclusions: This study demonstrates that alterations in the trajectory of neuronal growth throughout the lifespan are a core pathological features of idiopathic and syndromic autism. However, dup(15) causes persistent neuronal volume deficits in adolescence and adulthood, with prominent neuronal growth deficits in all major compartments of the limbic system. The more severe neuronal nuclear and cytoplasic volume deficits in syndromic autism found in this study and the more severe focal developmental defects in the limbic system in dup(15) previously reported in this cohort may contribute to the high prevalence of early onset intractable epilepsy and sudden unexpected death in epilepsy.
Figures
Similar articles
-
Neuronal nucleus and cytoplasm volume deficit in children with autism and volume increase in adolescents and adults.Acta Neuropathol Commun. 2015 Jan 17;3:2. doi: 10.1186/s40478-015-0183-5. Acta Neuropathol Commun. 2015. PMID: 25595448 Free PMC article.
-
The role of reduced expression of fragile X mental retardation protein in neurons and increased expression in astrocytes in idiopathic and syndromic autism (duplications 15q11.2-q13).Autism Res. 2018 Oct;11(10):1316-1331. doi: 10.1002/aur.2003. Epub 2018 Aug 14. Autism Res. 2018. PMID: 30107092
-
Brain-region-specific alterations of the trajectories of neuronal volume growth throughout the lifespan in autism.Acta Neuropathol Commun. 2014 Mar 10;2:28. doi: 10.1186/2051-5960-2-28. Acta Neuropathol Commun. 2014. PMID: 24612906 Free PMC article.
-
The 15q11.2 BP1-BP2 microdeletion syndrome: a review.Int J Mol Sci. 2015 Feb 13;16(2):4068-82. doi: 10.3390/ijms16024068. Int J Mol Sci. 2015. PMID: 25689425 Free PMC article. Review.
-
Autism and 15q11-q13 disorders: behavioral, genetic, and pathophysiological issues.Ment Retard Dev Disabil Res Rev. 2004;10(4):284-91. doi: 10.1002/mrdd.20042. Ment Retard Dev Disabil Res Rev. 2004. PMID: 15666333 Review.
Cited by
-
Neuronal volume of the hippocampal regions in ageing.J Anat. 2020 Aug;237(2):301-310. doi: 10.1111/joa.13189. Epub 2020 Apr 20. J Anat. 2020. PMID: 32314379 Free PMC article.
-
Abnormal sleep physiology in children with 15q11.2-13.1 duplication (Dup15q) syndrome.Mol Autism. 2021 Aug 3;12(1):54. doi: 10.1186/s13229-021-00460-8. Mol Autism. 2021. PMID: 34344470 Free PMC article.
-
The Protracted Maturation of Associative Layer IIIC Pyramidal Neurons in the Human Prefrontal Cortex During Childhood: A Major Role in Cognitive Development and Selective Alteration in Autism.Front Psychiatry. 2019 Mar 14;10:122. doi: 10.3389/fpsyt.2019.00122. eCollection 2019. Front Psychiatry. 2019. PMID: 30923504 Free PMC article.
-
Development and evaluation of an autism pig model.Lab Anim (NY). 2024 Dec;53(12):376-386. doi: 10.1038/s41684-024-01475-3. Epub 2024 Nov 12. Lab Anim (NY). 2024. PMID: 39533118 Free PMC article.
-
Single-cell analysis of dup15q syndrome reveals developmental and postnatal molecular changes in autism.Nat Commun. 2025 Jul 4;16(1):6177. doi: 10.1038/s41467-025-61184-4. Nat Commun. 2025. PMID: 40615364 Free PMC article.
References
Publication types
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

