Growth Hormone Ameliorates the Radiotherapy-Induced Ovarian Follicular Loss in Rats: Impact on Oxidative Stress, Apoptosis and IGF-1/IGF-1R Axis
- PMID: 26465611
- PMCID: PMC4605641
- DOI: 10.1371/journal.pone.0140055
Growth Hormone Ameliorates the Radiotherapy-Induced Ovarian Follicular Loss in Rats: Impact on Oxidative Stress, Apoptosis and IGF-1/IGF-1R Axis
Abstract
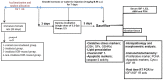
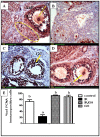
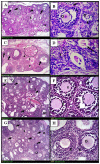
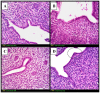
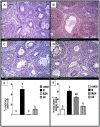
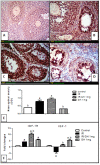
Radiotherapy is one of the standard cytotoxic therapies for cancer. However, it has a profound impact on ovarian function leading to premature ovarian failure and infertility. Since none of the currently available methods for fertility preservation guarantees future fertility, the need for an effective radioprotective agent is highly intensified. The present study investigated the mechanisms of the potential radioprotective effect of growth hormone (GH) on γ irradiation-induced ovarian failure and the impact of the insulin like growth factor 1 (IGF-1) in the underlying protection. Immature female Sprague-Dawley rats were either exposed to single whole body irradiation (3.2 Gy) and/or treated with GH (1 mg/kg s.c). Experimental γ-irradiation produced an array of ovarian dysfunction that was evident by assessment of hormonal changes, follicular development, proliferation marker (PCNA), oxidative stress as well as apoptotic markers. In addition, IGF-1/IGF-1R axis expression was assessed using real-time PCR and immunolocalization techniques. Furthermore, after full maturity, fertility assessment was performed. GH significantly enhanced follicular development and restored anti-Mullerian hormone serum level as compared with the irradiated group. In addition, GH significantly ameliorated the deleterious effects of irradiation on oxidative status, PCNA and apoptosis. Interestingly, GH was shown to enhance the ovarian IGF-1 at transcription and translation levels, a property that contributes significantly to its radioprotective effect. Finally, GH regained the fertility that was lost following irradiation. In conclusion, GH showed a radioprotective effect and rescued the ovarian reserve through increasing local IGF-1 level and counteracting the oxidative stress-mediated apoptosis.
Conflict of interest statement
Figures
References
-
- Steel GG. Basic clinical radiobiology (3rdedn.), Edward Arnold Publishers; London; 2002.
-
- Nair CK, Parida DK, Nomura T. Radioprotectors in radiotherapy. J Radiat Res. 2001; 42(1): 21–37. - PubMed
-
- Meirow D. Reproduction post-chemotherapy in young cancer patients. Mol Cell Endocrinol. 2000; 169, 123–131. - PubMed
-
- Pacey AA. Fertility issues in survivors from adolescent cancers. Cancer Treat Rev. 2007; 33:646–655. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

