Information processing in the hemisphere of the cerebellar cortex for control of wrist movement
- PMID: 26467515
- PMCID: PMC4760480
- DOI: 10.1152/jn.00530.2015
Information processing in the hemisphere of the cerebellar cortex for control of wrist movement
Abstract
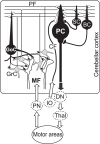
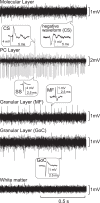
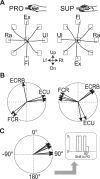
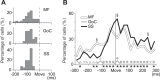
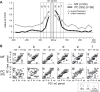
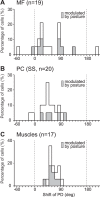
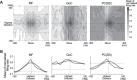
A region of cerebellar lobules V and VI makes strong loop connections with the primary motor (M1) and premotor (PM) cortical areas and is assumed to play essential roles in limb motor control. To examine its functional role, we compared the activities of its input, intermediate, and output elements, i.e., mossy fibers (MFs), Golgi cells (GoCs), and Purkinje cells (PCs), in three monkeys performing wrist movements in two different forearm postures. The results revealed distinct steps of information processing. First, MF activities displayed temporal and directional properties that were remarkably similar to those of M1/PM neurons, suggesting that MFs relay near copies of outputs from these motor areas. Second, all GoCs had a stereotyped pattern of activity independent of movement direction or forearm posture. Instead, GoC activity resembled an average of all MF activities. Therefore, inhibitory GoCs appear to provide a filtering function that passes only prominently modulated MF inputs to granule cells. Third, PCs displayed highly complex spatiotemporal patterns of activity, with coordinate frames distinct from those of MF inputs and directional tuning that changed abruptly before movement onset. The complexity of PC activities may reflect rapidly changing properties of the peripheral motor apparatus during movement. Overall, the cerebellar cortex appears to transform a representation of outputs from M1/PM into different movement representations in a posture-dependent manner and could work as part of a forward model that predicts the state of the peripheral motor apparatus.
Keywords: Golgi cell; Purkinje cell; cerebellar cortex; monkey; mossy fiber; motor control.
Copyright © 2016 the American Physiological Society.
Figures





Similar articles
-
Golgi cells operate as state-specific temporal filters at the input stage of the cerebellar cortex.J Neurosci. 2010 Dec 15;30(50):17004-14. doi: 10.1523/JNEUROSCI.3513-10.2010. J Neurosci. 2010. PMID: 21159970 Free PMC article.
-
Movement-related inputs to intermediate cerebellum of the monkey.J Neurophysiol. 1993 Jan;69(1):74-94. doi: 10.1152/jn.1993.69.1.74. J Neurophysiol. 1993. PMID: 8433135
-
Cerebellar cortical activity during antagonist cocontraction and reciprocal inhibition of forearm muscles.J Neurophysiol. 1984 Jan;51(1):32-49. doi: 10.1152/jn.1984.51.1.32. J Neurophysiol. 1984. PMID: 6693934
-
Detection of sequences in the cerebellar cortex: numerical estimate of the possible number of tidal-wave inducing sequences represented.J Physiol Paris. 2003 Jul-Nov;97(4-6):591-600. doi: 10.1016/j.jphysparis.2004.01.016. J Physiol Paris. 2003. PMID: 15242668 Review.
-
The role of calcium in synaptic plasticity and motor learning in the cerebellar cortex.Neurosci Biobehav Rev. 2012 Apr;36(4):1153-62. doi: 10.1016/j.neubiorev.2012.01.005. Epub 2012 Jan 28. Neurosci Biobehav Rev. 2012. PMID: 22305995 Review.
Cited by
-
Distinct representations of body and head motion are dynamically encoded by Purkinje cell populations in the macaque cerebellum.Elife. 2022 Apr 25;11:e75018. doi: 10.7554/eLife.75018. Elife. 2022. PMID: 35467528 Free PMC article.
-
Total sleep deprivation reduces the table tennis anticipation performance of young men: A functional magnetic resonance imaging study.iScience. 2023 Sep 19;26(10):107973. doi: 10.1016/j.isci.2023.107973. eCollection 2023 Oct 20. iScience. 2023. PMID: 37822501 Free PMC article.
-
Dynamics of directional tuning and reference frames in humans: A high-density EEG study.Sci Rep. 2018 May 29;8(1):8205. doi: 10.1038/s41598-018-26609-9. Sci Rep. 2018. PMID: 29844584 Free PMC article.
-
The Cerebro-Cerebellum as a Locus of Forward Model: A Review.Front Syst Neurosci. 2020 Apr 9;14:19. doi: 10.3389/fnsys.2020.00019. eCollection 2020. Front Syst Neurosci. 2020. PMID: 32327978 Free PMC article.
-
Dysmetria and Errors in Predictions: The Role of Internal Forward Model.Int J Mol Sci. 2020 Sep 20;21(18):6900. doi: 10.3390/ijms21186900. Int J Mol Sci. 2020. PMID: 32962256 Free PMC article. Review.
References
-
- Ambrosi G, Flace P, Lorusso L, Girolamo F, Rizzi A, Bosco L, Errede M, Virgintino D, Roncali L, Benagiano V. Non-traditional large neurons in the granular layer of the cerebellar cortex. Eur J Histochem 51, Suppl 1: 59–64, 2007. - PubMed
-
- Bourbonnais D, Krieger C, Smith AM. Cerebellar cortical activity during stretch of antagonist muscles. Can J Physiol Pharmacol 64: 1202–1213, 1986. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

