Gnb isoforms control a signaling pathway comprising Rac1, Plcβ2, and Plcβ3 leading to LFA-1 activation and neutrophil arrest in vivo
- PMID: 26468229
- PMCID: PMC4722285
- DOI: 10.1182/blood-2015-06-651034
Gnb isoforms control a signaling pathway comprising Rac1, Plcβ2, and Plcβ3 leading to LFA-1 activation and neutrophil arrest in vivo
Abstract
Chemokines are required for leukocyte recruitment and appropriate host defense and act through G protein-coupled receptors (GPCRs), which induce downstream signaling leading to integrin activation. Although the α and β subunits of the GPCRs are the first intracellular molecules that transduce signals after ligand binding and are therefore indispensable for downstream signaling, relatively little is known about their contribution to lymphocyte function-associated antigen 1 (LFA-1) activation and leukocyte recruitment. We used knockout mice and short hairpin RNA to knock down guanine nucleotide binding protein (GNB) isoforms (GNB1, GNB2, GNB4, and GNB5) in HL60 cells and primary murine hematopoietic cells. Neutrophil function was assessed by using intravital microscopy, flow chamber assays, and chemotaxis and biochemistry studies. We unexpectedly discovered that all expressed GNB isoforms are required for LFA-1 activation. Their downregulation led to a significant impairment of LFA-1 activation, which was demonstrated in vitro and in vivo. Furthermore, we showed that GPCR activation leads to Ras-related C3 botulinum toxin substrate 1 (Rac1)-dependent activation of both phospholipase C β2 (Plcβ2) and Plcβ3. They act nonredundantly to produce inositol triphosphate-mediated intracellular Ca(2+) flux and LFA-1 activation that support chemokine-induced arrest in vivo. In a complex inflammatory disease model, Plcβ2-, Plcβ3-, or Rac1-deficient mice were protected from lipopolysaccharide-induced lung injury. Taken together, we demonstrated that all Gnb isoforms are required for chemokine-induced downstream signaling, and Rac1, Plcβ2, and Plcβ3 are critically involved in integrin activation and leukocyte arrest.
© 2016 by The American Society of Hematology.
Figures
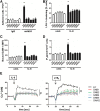

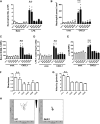
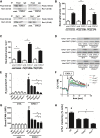
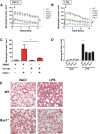
Comment in
-
Fine-tuning of integrin activation.Blood. 2016 Jan 21;127(3):275-6. doi: 10.1182/blood-2015-10-676908. Blood. 2016. PMID: 26796106 No abstract available.
Similar articles
-
HS1 deficiency impairs neutrophil recruitment in vivo and activation of the small GTPases Rac1 and Rap1.J Leukoc Biol. 2017 May;101(5):1133-1142. doi: 10.1189/jlb.1A0416-195R. Epub 2017 Jan 25. J Leukoc Biol. 2017. PMID: 28122813
-
Fine-tuning of integrin activation.Blood. 2016 Jan 21;127(3):275-6. doi: 10.1182/blood-2015-10-676908. Blood. 2016. PMID: 26796106 No abstract available.
-
Synergistic Ca2+ responses by G{alpha}i- and G{alpha}q-coupled G-protein-coupled receptors require a single PLC{beta} isoform that is sensitive to both G{beta}{gamma} and G{alpha}q.J Biol Chem. 2011 Jan 14;286(2):942-51. doi: 10.1074/jbc.M110.198200. Epub 2010 Oct 29. J Biol Chem. 2011. PMID: 21036901 Free PMC article.
-
T cell LFA-1 engagement induces HuR-dependent cytokine mRNA stabilization through a Vav-1, Rac1/2, p38MAPK and MKK3 signaling cascade.PLoS One. 2010 Dec 29;5(12):e14450. doi: 10.1371/journal.pone.0014450. PLoS One. 2010. PMID: 21206905 Free PMC article.
-
The correlation between multidomain enzymes and multiple activation mechanisms--the case of phospholipase Cβ and its membrane interactions.Biochim Biophys Acta. 2011 Dec;1808(12):2940-7. doi: 10.1016/j.bbamem.2011.08.028. Epub 2011 Aug 30. Biochim Biophys Acta. 2011. PMID: 21906583 Free PMC article. Review.
Cited by
-
A multidimensional recommendation framework for identifying biological targets to aid the diagnosis and treatment of liver metastasis in patients with colorectal cancer.Mol Cancer. 2024 Oct 24;23(1):239. doi: 10.1186/s12943-024-02155-z. Mol Cancer. 2024. PMID: 39449040 Free PMC article.
-
β2 Integrins-Multi-Functional Leukocyte Receptors in Health and Disease.Int J Mol Sci. 2020 Feb 19;21(4):1402. doi: 10.3390/ijms21041402. Int J Mol Sci. 2020. PMID: 32092981 Free PMC article. Review.
-
A suppressive role of guanine nucleotide-binding protein subunit beta-4 inhibited by DNA methylation in the growth of anti-estrogen resistant breast cancer cells.BMC Cancer. 2018 Aug 13;18(1):817. doi: 10.1186/s12885-018-4711-0. BMC Cancer. 2018. PMID: 30103729 Free PMC article.
-
Nexinhib20 Inhibits Neutrophil Adhesion and β2 Integrin Activation by Antagonizing Rac-1-Guanosine 5'-Triphosphate Interaction.J Immunol. 2022 Oct 15;209(8):1574-1585. doi: 10.4049/jimmunol.2101112. Epub 2022 Sep 7. J Immunol. 2022. PMID: 36165184 Free PMC article.
-
β2 integrin activation and signal transduction in leukocyte recruitment.Am J Physiol Cell Physiol. 2021 Aug 1;321(2):C308-C316. doi: 10.1152/ajpcell.00560.2020. Epub 2021 Jun 16. Am J Physiol Cell Physiol. 2021. PMID: 34133240 Free PMC article. Review.
References
-
- Ley K, Laudanna C, Cybulsky MI, Nourshargh S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol. 2007;7(9):678–689. - PubMed
-
- Herter J, Zarbock A. Integrin Regulation during Leukocyte Recruitment. J Immunol. 2013;190(9):4451–4457. - PubMed
-
- Takagi J, Petre BM, Walz T, Springer TA. Global conformational rearrangements in integrin extracellular domains in outside-in and inside-out signaling. Cell. 2002;110(5):599–611. - PubMed
-
- Zarbock A, Kempf T, Wollert KC, Vestweber D. Leukocyte integrin activation and deactivation: novel mechanisms of balancing inflammation. J Mol Med (Berl) 2012;90(4):353–359. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

