Conditional deletion of caspase-8 in macrophages alters macrophage activation in a RIPK-dependent manner
- PMID: 26471282
- PMCID: PMC4608154
- DOI: 10.1186/s13075-015-0794-z
Conditional deletion of caspase-8 in macrophages alters macrophage activation in a RIPK-dependent manner
Abstract
Introduction: Although caspase-8 is a well-established initiator of apoptosis and suppressor of necroptosis, recent evidence suggests that this enzyme maintains functions beyond its role in cell death. As cells of the innate immune system, and in particular macrophages, are now at the forefront of autoimmune disease pathogenesis, we examined the potential involvement of caspase-8 within this population.
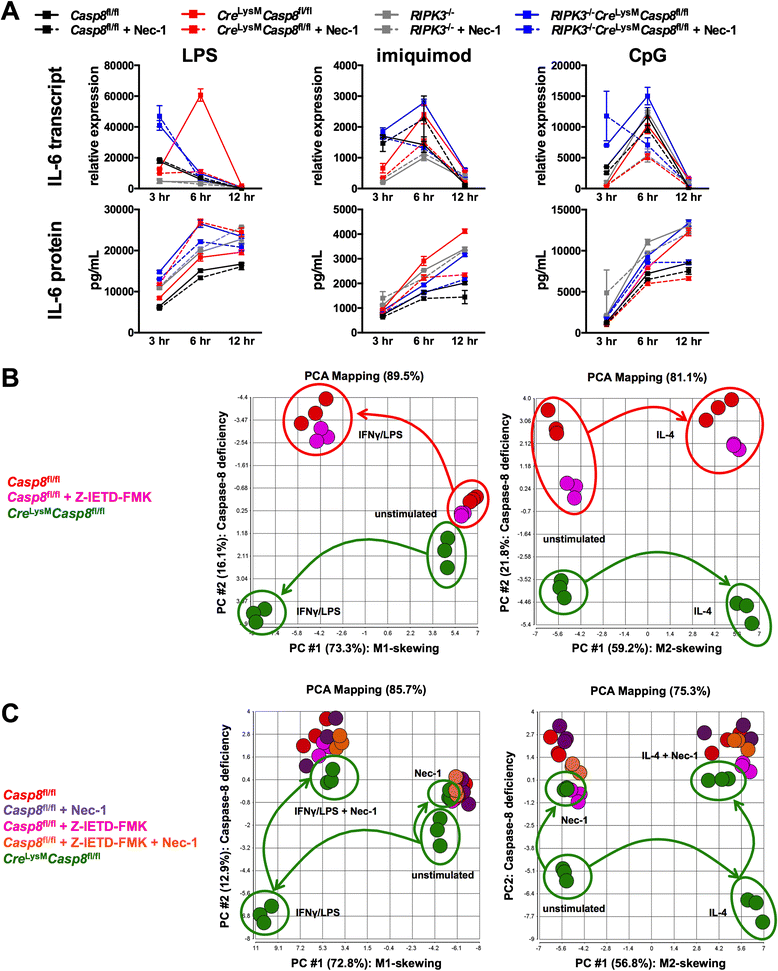
Methods: Cre (LysM) Casp8 (fl/fl) mice were bred via a cross between Casp8 (fl/fl) mice and Cre (LysM) mice, and RIPK3 (-/-) Cre (LysM) Casp8 (fl/fl) mice were generated to assess the contribution of receptor-interacting serine-threonine kinase (RIPK)3. Immunohistochemical and immunofluorescence analyses were used to examine renal damage. Flow cytometric analysis was employed to characterize splenocyte distribution and activation. Cre (LysM) Casp8 (fl/fl) mice were treated with either Toll-like receptor (TLR) agonists or oral antibiotics to assess their response to TLR activation or TLR agonist removal. Luminex-based assays and enzyme-linked immunosorbent assays were used to measure cytokine/chemokine and immunoglobulin levels in serum and cytokine levels in cell culture studies. In vitro cell culture was used to assess macrophage response to cell death stimuli, TLR activation, and M1/M2 polarization. Data were compared using the Mann-Whitney U test.
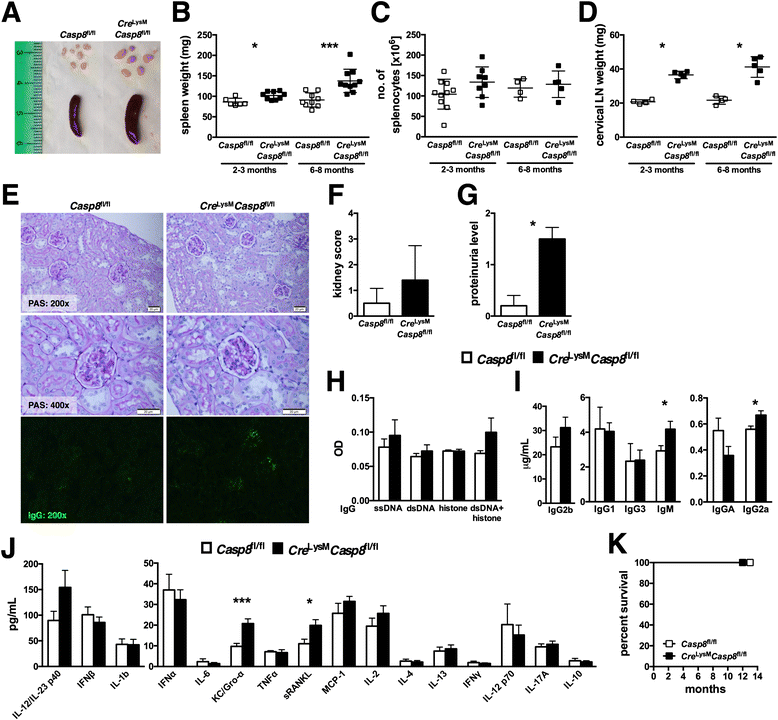
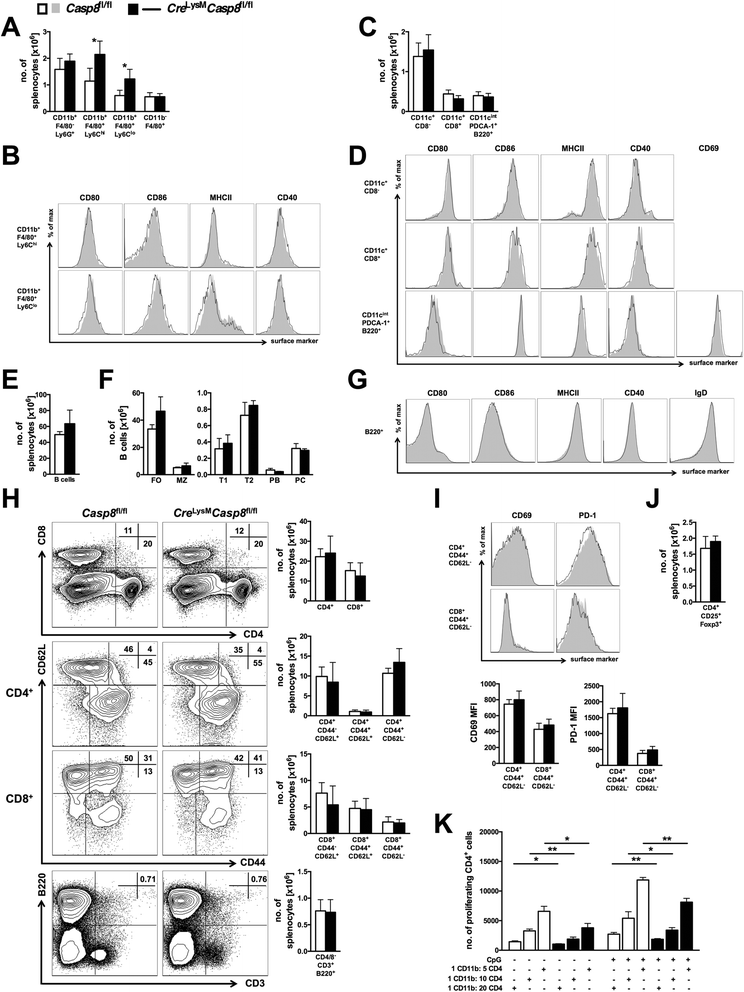
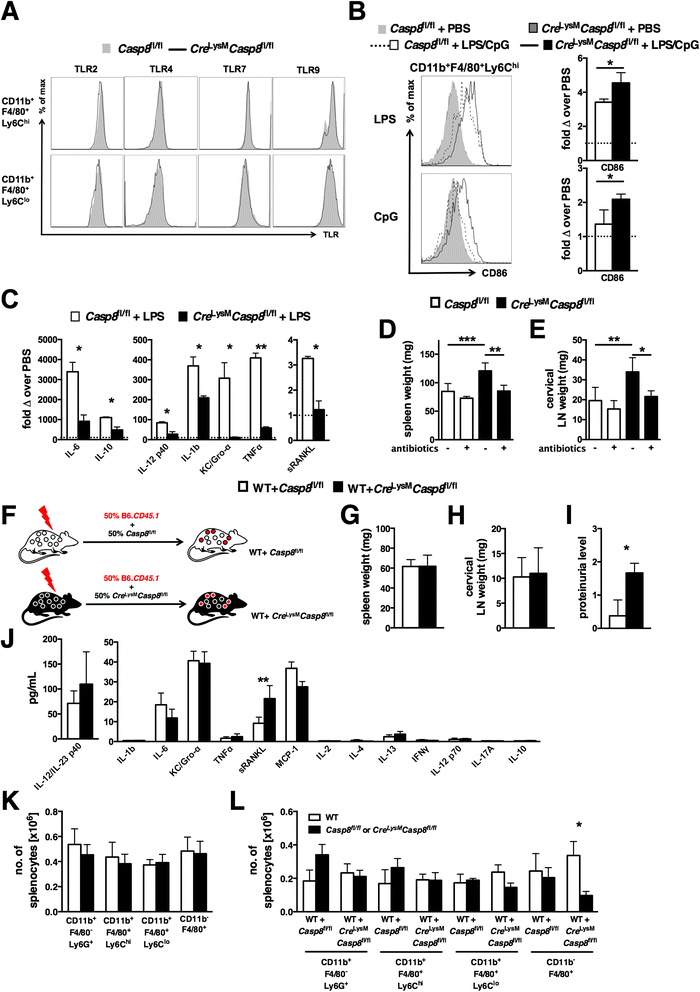
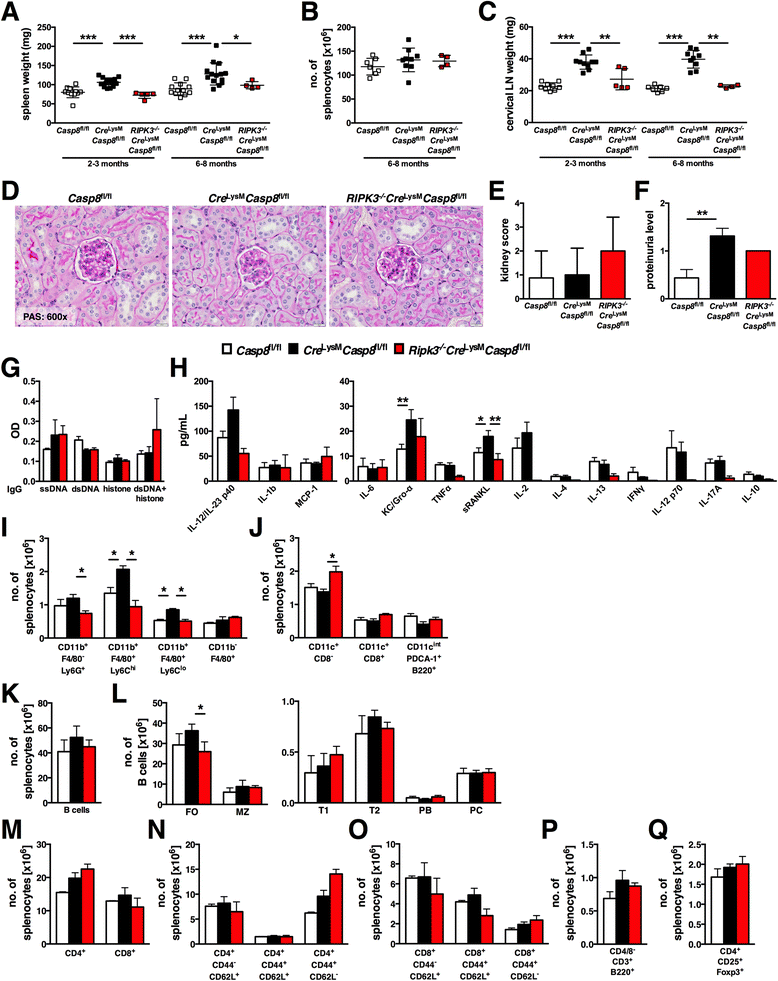
Results: Loss of caspase-8 expression in macrophages promotes onset of a mild systemic inflammatory disease, which is preventable by the deletion of RIPK3. In vitro cell culture studies reveal that caspase-8-deficient macrophages are prone to a caspase-independent death in response to death receptor ligation; yet, caspase-8-deficient macrophages are not predisposed to unchecked survival, as analysis of mixed bone marrow chimeric mice demonstrates that caspase-8 deficiency does not confer preferential expansion of myeloid populations. Loss of caspase-8 in macrophages dictates the response to TLR activation, as injection of TLR ligands upregulates expression of costimulatory CD86 on the Ly6C(high)CD11b(+)F4/80(+) splenic cells, and oral antibiotic treatment to remove microbiota prevents splenomegaly and lymphadenopathy in Cre (LysM) Casp8 (fl/fl) mice. Further, caspase-8-deficient macrophages are hyperresponsive to TLR activation and exhibit aberrant M1 macrophage polarization due to RIPK activity.
Conclusions: These data demonstrate that caspase-8 functions uniquely in macrophages by controlling the response to TLR activation and macrophage polarization in an RIPK-dependent manner.
Figures
Similar articles
-
The caspase-8/RIPK3 signaling axis in antigen presenting cells controls the inflammatory arthritic response.Arthritis Res Ther. 2017 Oct 4;19(1):224. doi: 10.1186/s13075-017-1436-4. Arthritis Res Ther. 2017. PMID: 28978351 Free PMC article.
-
XIAP Loss Triggers RIPK3- and Caspase-8-Driven IL-1β Activation and Cell Death as a Consequence of TLR-MyD88-Induced cIAP1-TRAF2 Degradation.Cell Rep. 2017 Jul 18;20(3):668-682. doi: 10.1016/j.celrep.2017.06.073. Cell Rep. 2017. PMID: 28723569
-
Remarkably Robust Antiviral Immune Response despite Combined Deficiency in Caspase-8 and RIPK3.J Immunol. 2018 Oct 15;201(8):2244-2255. doi: 10.4049/jimmunol.1800110. Epub 2018 Sep 7. J Immunol. 2018. PMID: 30194111 Free PMC article.
-
Forkhead Box O1 Regulates Macrophage Polarization Following Staphylococcus aureus Infection: Experimental Murine Data and Review of the Literature.Clin Rev Allergy Immunol. 2016 Dec;51(3):353-369. doi: 10.1007/s12016-016-8531-1. Clin Rev Allergy Immunol. 2016. PMID: 26924010 Review.
-
Necroptosis and Inflammation.Annu Rev Biochem. 2016 Jun 2;85:743-63. doi: 10.1146/annurev-biochem-060815-014830. Epub 2016 Feb 8. Annu Rev Biochem. 2016. PMID: 26865533 Review.
Cited by
-
The inflammatory role of phagocyte apoptotic pathways in rheumatic diseases.Nat Rev Rheumatol. 2016 Aug 23;12(9):543-58. doi: 10.1038/nrrheum.2016.132. Nat Rev Rheumatol. 2016. PMID: 27549026 Free PMC article. Review.
-
Targeting caspase-8: a new strategy for combating hepatocellular carcinoma.Front Immunol. 2024 Dec 12;15:1501659. doi: 10.3389/fimmu.2024.1501659. eCollection 2024. Front Immunol. 2024. PMID: 39726605 Free PMC article. Review.
-
Apoptotic cell death in disease-Current understanding of the NCCD 2023.Cell Death Differ. 2023 May;30(5):1097-1154. doi: 10.1038/s41418-023-01153-w. Epub 2023 Apr 26. Cell Death Differ. 2023. PMID: 37100955 Free PMC article. Review.
-
The intersection of cell death and inflammasome activation.Cell Mol Life Sci. 2016 Jun;73(11-12):2349-67. doi: 10.1007/s00018-016-2205-2. Epub 2016 Apr 11. Cell Mol Life Sci. 2016. PMID: 27066895 Free PMC article. Review.
-
Bim suppresses the development of SLE by limiting myeloid inflammatory responses.J Exp Med. 2017 Dec 4;214(12):3753-3773. doi: 10.1084/jem.20170479. Epub 2017 Nov 7. J Exp Med. 2017. PMID: 29114065 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- AI099009/AI/NIAID NIH HHS/United States
- GM071723/GM/NIGMS NIH HHS/United States
- AR054796/AR/NIAMS NIH HHS/United States
- P01 HL108795/HL/NHLBI NIH HHS/United States
- HL108795/HL/NHLBI NIH HHS/United States
- I01 BX000201/BX/BLRD VA/United States
- P01 AG049665/AG/NIA NIH HHS/United States
- R01 GM071723/GM/NIGMS NIH HHS/United States
- AR050250/AR/NIAMS NIH HHS/United States
- AR060169/AR/NIAMS NIH HHS/United States
- R01 AI099009/AI/NIAID NIH HHS/United States
- K01 AR066739/AR/NIAMS NIH HHS/United States
- R01 AR064349/AR/NIAMS NIH HHS/United States
- AR064349/AR/NIAMS NIH HHS/United States
- R01 AR050250/AR/NIAMS NIH HHS/United States
- R01 AR054796/AR/NIAMS NIH HHS/United States
- F32 AR060169/AR/NIAMS NIH HHS/United States
- K01 AR064313/AR/NIAMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

