The cardiac repair benefits of inflammation do not persist: evidence from mast cell implantation
- PMID: 26471858
- PMCID: PMC4687709
- DOI: 10.1111/jcmm.12703
The cardiac repair benefits of inflammation do not persist: evidence from mast cell implantation
Abstract
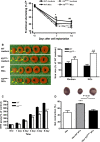
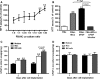
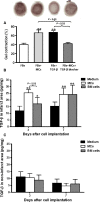
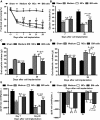
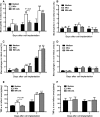
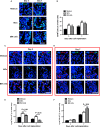
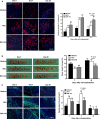
Multiple mechanisms contribute to progressive cardiac dysfunction after myocardial infarction (MI) and inflammation is an important mediator. Mast cells (MCs) trigger inflammation after MI by releasing bio-active factors that contribute to healing. c-Kit-deficient (Kit(W/W-v) ) mice have dysfunctional MCs and develop severe ventricular dilatation post-MI. We explored the role of MCs in post-MI repair. Mouse wild-type (WT) and Kit(W/W-v) MCs were obtained from bone marrow (BM). MC effects on fibroblasts were examined in vitro by proliferation and gel contraction assays. MCs were implanted into infarcted mouse hearts and their effects were evaluated using molecular, cellular and cardiac functional analyses. In contrast to WT, Kit(W/W-v) MC transplantation into Kit(W/W-v) mice did not improve cardiac function or scar size post-MI. Kit(W/W-v) MCs induced significantly reduced fibroblast proliferation and contraction compared to WT MCs. MC influence on fibroblast proliferation was Basic fibroblast growth factor (bFGF)-dependent and MC-induced fibroblast contractility functioned through transforming growth factor (TGF)-β. WT MCs transiently rescue cardiac function early post-MI, but the benefits of BM cell implantation lasted longer. MCs induced increased inflammation compared to the BM-injected mice, with increased neutrophil infiltration and infarct tumour necrosis factor-α (TNF-α) concentration. This augmented inflammation was followed by increased angiogenesis and myofibroblast formation and reduced scar size at early time-points. Similar to the functional data, these beneficial effects were transient, largely vanishing by day 28. Dysfunctional Kit(W/W-v) MCs were unable to rescue cardiac function post-MI. WT MC implantation transiently enhanced angiogenesis and cardiac function. These data suggest that increased inflammation is beneficial to cardiac repair, but these effects are not persistent.
Keywords: c-Kit-deficient mice; cell transplantation; inflammatory response; mast cells; myocardial infarction; myofibroblasts.
© 2015 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures
Similar articles
-
Mast cells regulate myofilament calcium sensitization and heart function after myocardial infarction.J Exp Med. 2016 Jun 27;213(7):1353-74. doi: 10.1084/jem.20160081. J Exp Med. 2016. PMID: 27353089 Free PMC article.
-
Stem cell factor/mast cell/CCL2/monocyte/macrophage axis promotes Coxsackievirus B3 myocarditis and cardiac fibrosis by increasing Ly6Chigh monocyte influx and fibrogenic mediators production.Immunology. 2022 Dec;167(4):590-605. doi: 10.1111/imm.13556. Epub 2022 Aug 23. Immunology. 2022. PMID: 36054617
-
Mast Cells Infiltrating Inflamed or Transformed Gut Alternatively Sustain Mucosal Healing or Tumor Growth.Cancer Res. 2015 Sep 15;75(18):3760-70. doi: 10.1158/0008-5472.CAN-14-3767. Epub 2015 Jul 23. Cancer Res. 2015. PMID: 26206557
-
Fibroblasts in myocardial infarction: a role in inflammation and repair.J Mol Cell Cardiol. 2014 May;70:74-82. doi: 10.1016/j.yjmcc.2013.11.015. Epub 2013 Dec 7. J Mol Cell Cardiol. 2014. PMID: 24321195 Free PMC article. Review.
-
Properties and Functions of Fibroblasts and Myofibroblasts in Myocardial Infarction.Cells. 2022 Apr 20;11(9):1386. doi: 10.3390/cells11091386. Cells. 2022. PMID: 35563692 Free PMC article. Review.
Cited by
-
Histological Evidence for Therapeutic Induction of Angiogenesis Using Mast Cells and Platelet-Rich Plasma within A Bioengineered Scaffold following Rat Hindlimb Ischemia.Cell J. 2020 Jan;21(4):391-400. doi: 10.22074/cellj.2020.6287. Epub 2019 Jul 29. Cell J. 2020. PMID: 31376320 Free PMC article.
-
Mast Cells in Liver Fibrogenesis.Cells. 2019 Nov 13;8(11):1429. doi: 10.3390/cells8111429. Cells. 2019. PMID: 31766207 Free PMC article. Review.
-
Genetic Deficiency and Pharmacological Stabilization of Mast Cells Ameliorate Pressure Overload-Induced Maladaptive Right Ventricular Remodeling in Mice.Int J Mol Sci. 2020 Nov 30;21(23):9099. doi: 10.3390/ijms21239099. Int J Mol Sci. 2020. PMID: 33265921 Free PMC article.
-
The Immune System, An Arrow into the Heart. Principles of Cardioimmunology, An Emerging Branch in Medicine.Curr Vasc Pharmacol. 2025;23(3):162-171. doi: 10.2174/0115701611325234241202073459. Curr Vasc Pharmacol. 2025. PMID: 39810536 Review.
-
Cardiac fibrogenesis: an immuno-metabolic perspective.Front Physiol. 2024 Mar 21;15:1336551. doi: 10.3389/fphys.2024.1336551. eCollection 2024. Front Physiol. 2024. PMID: 38577624 Free PMC article. Review.
References
-
- Batlle M, Pérez‐Villa F, Lazaro A, et al Correlation between mast cell density and myocardial fibrosis in congestive heart failure patients. Transplant Proc. 2007; 39: 2347–9. - PubMed
-
- Engels W, Reiters PH, Daemen MJ, et al Transmural changes in mast cell density in rat heart after infarct induction in vivo . J Pathol. 1995; 177: 423–9. - PubMed
-
- Frangogiannis NG, Lindsey ML, Michael LH, et al Resident cardiac mast cells degranulate and release preformed TNF‐alpha, initiating the cytokine cascade in experimental canine myocardial ischemia/reperfusion. Circulation. 1998; 98: 699–710. - PubMed
-
- de Almeida A, Mustin D, Forman MF, et al Effects of mast cells on the behavior of isolated heart fibroblasts: modulation of collagen remodeling and gene expression. J Cell Physiol. 2002; 191: 51–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

