Regulation of mechanosensitive biliary epithelial transport by the epithelial Na(+) channel
- PMID: 26475057
- PMCID: PMC4780683
- DOI: 10.1002/hep.28301
Regulation of mechanosensitive biliary epithelial transport by the epithelial Na(+) channel
Abstract
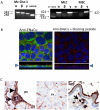
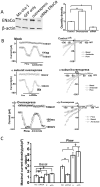
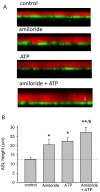
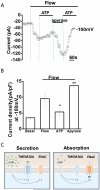
Intrahepatic biliary epithelial cells (BECs), also known as cholangiocytes, modulate the volume and composition of bile through the regulation of secretion and absorption. While mechanosensitive Cl(-) efflux has been identified as an important secretory pathway, the counterabsorptive pathways have not been identified. In other epithelial cells, the epithelial Na(+) channel (ENaC) has been identified as an important contributor to fluid absorption; however, its expression and function in BECs have not been previously studied. Our studies revealed the presence of α, β, and γ ENaC subunits in human BECs and α and γ subunits in mouse BECs. In studies of confluent mouse BEC monolayers, the ENaC contributes to the volume of surface fluid at the apical membrane during constitutive conditions. Further, functional studies using whole-cell patch clamp of single BECs demonstrated small constitutive Na(+) currents, which increased significantly in response to fluid-flow or shear. The magnitude of Na(+) currents was proportional to the shear force, displayed inward rectification and a reversal potential of +40 mV (ENa+ = +60 mV), and were abolished with removal of extracellular Na(+) (N-methyl-d-glucamine) or in the presence of amiloride. Transfection with ENaCα small interfering RNA significantly inhibited flow-stimulated Na(+) currents, while overexpression of the α subunit significantly increased currents. ENaC-mediated currents were positively regulated by proteases and negatively regulated by extracellular adenosine triphosphate.
Conclusion: These studies represent the initial characterization of mechanosensitive Na(+) currents activated by flow in biliary epithelium; understanding the role of mechanosensitive transport pathways may provide strategies to modulate the volume and composition of bile during cholestatic conditions. (Hepatology 2016;63:538-549).
© 2015 by the American Association for the Study of Liver Diseases.
Figures


Similar articles
-
Mechanosensor transient receptor potential vanilloid member 4 (TRPV4) regulates mouse cholangiocyte secretion and bile formation.Am J Physiol Gastrointest Liver Physiol. 2020 Feb 1;318(2):G277-G287. doi: 10.1152/ajpgi.00176.2019. Epub 2019 Nov 25. Am J Physiol Gastrointest Liver Physiol. 2020. PMID: 31760763 Free PMC article.
-
Identification and functional characterization of TMEM16A, a Ca2+-activated Cl- channel activated by extracellular nucleotides, in biliary epithelium.J Biol Chem. 2011 Jan 7;286(1):766-76. doi: 10.1074/jbc.M110.164970. Epub 2010 Nov 1. J Biol Chem. 2011. PMID: 21041307 Free PMC article.
-
PKCα regulates TMEM16A-mediated Cl⁻ secretion in human biliary cells.Am J Physiol Gastrointest Liver Physiol. 2016 Jan 1;310(1):G34-42. doi: 10.1152/ajpgi.00146.2015. Epub 2015 Nov 5. Am J Physiol Gastrointest Liver Physiol. 2016. PMID: 26542395 Free PMC article.
-
Regulation of epithelial sodium channels by the ubiquitin-proteasome proteolytic pathway.Am J Physiol Renal Physiol. 2006 Jun;290(6):F1285-94. doi: 10.1152/ajprenal.00432.2005. Am J Physiol Renal Physiol. 2006. PMID: 16682484 Review.
-
ENaC is inhibited by an increase in the intracellular Cl(-) concentration mediated through activation of Cl(-) channels.Pflugers Arch. 2003 Jan;445(4):504-12. doi: 10.1007/s00424-002-0958-y. Epub 2002 Nov 20. Pflugers Arch. 2003. PMID: 12548397 Review.
Cited by
-
Bile acids regulate the epithelial Na+ channel in native tissues through direct binding at multiple sites.J Physiol. 2022 Nov;600(21):4695-4711. doi: 10.1113/JP283318. Epub 2022 Sep 23. J Physiol. 2022. PMID: 36071685 Free PMC article.
-
Bile acids stimulate cholangiocyte fluid secretion by activation of transmembrane member 16A Cl- channels.Hepatology. 2018 Jul;68(1):187-199. doi: 10.1002/hep.29804. Epub 2018 May 9. Hepatology. 2018. PMID: 29360145 Free PMC article.
-
Epithelial Na + Channels Function as Extracellular Sensors.Compr Physiol. 2024 Mar 29;14(2):1-41. doi: 10.1002/cphy.c230015. Compr Physiol. 2024. PMID: 39109974 Free PMC article. Review.
-
Contributions of bile acids to gastrointestinal physiology as receptor agonists and modifiers of ion channels.Am J Physiol Gastrointest Liver Physiol. 2022 Feb 1;322(2):G201-G222. doi: 10.1152/ajpgi.00125.2021. Epub 2021 Nov 10. Am J Physiol Gastrointest Liver Physiol. 2022. PMID: 34755536 Free PMC article.
-
Murine epithelial sodium (Na+) channel regulation by biliary factors.J Biol Chem. 2019 Jun 28;294(26):10182-10193. doi: 10.1074/jbc.RA119.007394. Epub 2019 May 15. J Biol Chem. 2019. PMID: 31092599 Free PMC article.
References
-
- Fitz JG. Cellular mechanisms of bile secretion. In: Zakim D, Boyer TD, editors. Hepatology. 3 W.B. Saunders Company; Philadelphia: 1996. pp. 362–376.
-
- Roman RM, Feranchak AP, Salter KD, Wang Y, Fitz JG. Endogenous ATP regulates Cl− secretion in cultured human and rat biliary epithelial cells. Am J Physiol. 1999;276:G1391–G1400. - PubMed
-
- Fiorotto R, Spirli C, Fabris L, Cadamuro M, Okolicsanyi L, Strazzabosco M. Ursodeoxycholic acid stimulates cholangiocyte fluid secretion in mice via CFTR-dependent ATP secretion. Gastroenterology. 2007 Nov;133(5):1603–1613. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

