Developmental changes in the morphology of mouse hypoglossal motor neurons
- PMID: 26476929
- PMCID: PMC5009180
- DOI: 10.1007/s00429-015-1130-8
Developmental changes in the morphology of mouse hypoglossal motor neurons
Abstract
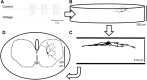
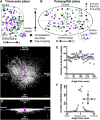
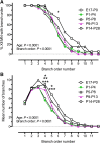
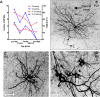
Hypoglossal motor neurons (XII MNs) innervate tongue muscles important in breathing, suckling and vocalization. Morphological properties of 103 XII MNs were studied using Neurobiotin™ filling in transverse brainstem slices from C57/Bl6 mice (n = 34) from embryonic day (E) 17 to postnatal day (P) 28. XII MNs from areas thought to innervate different tongue muscles showed similar morphology in most, but not all, features. Morphological properties of XII MNs were established prior to birth, not differing between E17-18 and P0. MN somatic volume gradually increased for the first 2 weeks post-birth. The complexity of dendritic branching and dendrite length of XII MNs increased throughout development (E17-P28). MNs in the ventromedial XII motor nucleus, likely to innervate the genioglossus, frequently (42 %) had dendrites crossing to the contralateral side at all ages, but their number declined with postnatal development. Unexpectedly, putative dendritic spines were found in all XII MNs at all ages, and were primarily localized to XII MN somata and primary dendrites at E18-P4, increased in distal dendrites by P5-P8, and were later predominantly found in distal dendrites. Dye-coupling between XII MNs was common from E18 to P7, but declined strongly with maturation after P7. Axon collaterals were found in 20 % (6 of 28) of XII MNs with filled axons; collaterals terminated widely outside and, in one case, within the XII motor nucleus. These results reveal new morphological features of mouse XII MNs, and suggest that dendritic projection patterns, spine density and distribution, and dye-coupling patterns show specific developmental changes in mice.
Keywords: Axon collateral; Dendritic morphology; Dye-coupling; Postnatal development; Respiration; Spine.
Conflict of interest statement
All authors declare that there are no known or potential conflict of interest including any financial, personal or other relationships with other people or organizations within three years of beginning the submitted work that could inappropriately influence, or be perceived to influence, their work. Support and grant information Funded by the Australian National Health and Medical Research Council project Grants 569827 and APP1065884 (MCB, PGN), and 569680 (PGN), the Motor Neurone Disease Research Institute of Australia (MCB, PGN), the Australian Brain Foundation (MCB) and the Clive and Vera Ramaciotti Foundation (MCB). MJF was supported by an Australian Postgraduate Award. Funding bodies had no involvement in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit this article for publication.
Figures







Similar articles
-
Changes in the Morphology of Hypoglossal Motor Neurons in the Brainstem of Developing Rats.Anat Rec (Hoboken). 2019 Jun;302(6):869-892. doi: 10.1002/ar.23971. Epub 2018 Nov 13. Anat Rec (Hoboken). 2019. PMID: 30312533
-
Size-dependent dendritic maladaptations of hypoglossal motor neurons in SOD1G93A mice.Anat Rec (Hoboken). 2021 Jul;304(7):1562-1581. doi: 10.1002/ar.24542. Epub 2020 Nov 2. Anat Rec (Hoboken). 2021. PMID: 33099869
-
Motor Areas Show Altered Dendritic Structure in an Amyotrophic Lateral Sclerosis Mouse Model.Front Neurosci. 2017 Nov 1;11:609. doi: 10.3389/fnins.2017.00609. eCollection 2017. Front Neurosci. 2017. PMID: 29163013 Free PMC article.
-
Noradrenergic modulation of hypoglossal motoneuron excitability: developmental and putative state-dependent mechanisms.Arch Ital Biol. 2011 Dec 1;149(4):426-53. doi: 10.4449/aib.v149i4.1271. Arch Ital Biol. 2011. PMID: 22205594 Review.
-
Gene delivery to the hypoglossal motor system: preclinical studies and translational potential.Gene Ther. 2021 Aug;28(7-8):402-412. doi: 10.1038/s41434-021-00225-1. Epub 2021 Feb 11. Gene Ther. 2021. PMID: 33574581 Free PMC article. Review.
Cited by
-
Sarcopenia of the longitudinal tongue muscles in rats.Respir Physiol Neurobiol. 2024 Jan;319:104180. doi: 10.1016/j.resp.2023.104180. Epub 2023 Oct 18. Respir Physiol Neurobiol. 2024. PMID: 37863156 Free PMC article.
-
Phrenic motor neuron loss in aged rats.J Neurophysiol. 2018 May 1;119(5):1852-1862. doi: 10.1152/jn.00868.2017. Epub 2018 Feb 7. J Neurophysiol. 2018. PMID: 29412773 Free PMC article.
-
Muscle-specific deletion of the vitamin D receptor in mice is associated with diaphragm muscle weakness.J Appl Physiol (1985). 2021 Jul 1;131(1):95-106. doi: 10.1152/japplphysiol.00194.2021. Epub 2021 May 20. J Appl Physiol (1985). 2021. PMID: 34013750 Free PMC article.
-
Cortical synaptic and dendritic spine abnormalities in a presymptomatic TDP-43 model of amyotrophic lateral sclerosis.Sci Rep. 2016 Nov 29;6:37968. doi: 10.1038/srep37968. Sci Rep. 2016. PMID: 27897242 Free PMC article.
-
C. elegans neurons have functional dendritic spines.Elife. 2019 Oct 4;8:e47918. doi: 10.7554/eLife.47918. Elife. 2019. PMID: 31584430 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

