Inhibition of mechanical allodynia in neuropathic pain by TLR5-mediated A-fiber blockade
- PMID: 26479925
- PMCID: PMC4752254
- DOI: 10.1038/nm.3978
Inhibition of mechanical allodynia in neuropathic pain by TLR5-mediated A-fiber blockade
Abstract
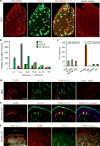
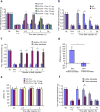
Mechanical allodynia, induced by normally innocuous low-threshold mechanical stimulation, represents a cardinal feature of neuropathic pain. Blockade or ablation of high-threshold, small-diameter unmyelinated group C nerve fibers (C-fibers) has limited effects on mechanical allodynia. Although large, myelinated group A fibers, in particular Aβ-fibers, have previously been implicated in mechanical allodynia, an A-fiber-selective pharmacological blocker is still lacking. Here we report a new method for targeted silencing of A-fibers in neuropathic pain. We found that Toll-like receptor 5 (TLR5) is co-expressed with neurofilament-200 in large-diameter A-fiber neurons in the dorsal root ganglion (DRG). Activation of TLR5 with its ligand flagellin results in neuronal entry of the membrane-impermeable lidocaine derivative QX-314, leading to TLR5-dependent blockade of sodium currents, predominantly in A-fiber neurons of mouse DRGs. Intraplantar co-application of flagellin and QX-314 (flagellin/QX-314) dose-dependently suppresses mechanical allodynia after chemotherapy, nerve injury, and diabetic neuropathy, but this blockade is abrogated in Tlr5-deficient mice. In vivo electrophysiology demonstrated that co-application of flagellin/QX-314 selectively suppressed Aβ-fiber conduction in naive and chemotherapy-treated mice. TLR5-mediated Aβ-fiber blockade, but not capsaicin-mediated C-fiber blockade, also reduced chemotherapy-induced ongoing pain without impairing motor function. Finally, flagellin/QX-314 co-application suppressed sodium currents in large-diameter human DRG neurons. Thus, our findings provide a new tool for targeted silencing of Aβ-fibers and neuropathic pain treatment.
Conflict of interest statement
All the authors have no financial interests in this study.
Figures


Comment in
-
Pain: TLR5 opens the door to neuropathic-pain treatment.Nat Rev Drug Discov. 2015 Dec;14(12):818. doi: 10.1038/nrd4785. Nat Rev Drug Discov. 2015. PMID: 26620409 No abstract available.
Similar articles
-
Pain: TLR5 opens the door to neuropathic-pain treatment.Nat Rev Drug Discov. 2015 Dec;14(12):818. doi: 10.1038/nrd4785. Nat Rev Drug Discov. 2015. PMID: 26620409 No abstract available.
-
Hypersensitivity of myelinated A-fibers via toll-like receptor 5 promotes mechanical allodynia in tenascin-X-deficient mice associated with Ehlers-Danlos syndrome.Sci Rep. 2023 Oct 28;13(1):18490. doi: 10.1038/s41598-023-45638-7. Sci Rep. 2023. PMID: 37898719 Free PMC article.
-
Targeting Toll-like receptors to treat chronic pain.Nat Med. 2015 Nov;21(11):1251-2. doi: 10.1038/nm.3986. Nat Med. 2015. PMID: 26540386 No abstract available.
-
New approach for investigating neuropathic allodynia by optogenetics.Pain. 2019 May;160 Suppl 1:S53-S58. doi: 10.1097/j.pain.0000000000001506. Pain. 2019. PMID: 31008850 Review.
-
Neuropathic Pain: Sensory Nerve Injury or Motor Nerve Injury?Adv Exp Med Biol. 2016;904:59-75. doi: 10.1007/978-94-017-7537-3_5. Adv Exp Med Biol. 2016. PMID: 26900063 Review.
Cited by
-
Peripheral neuroimmune interactions: selected review and some clinical implications.Clin Auton Res. 2021 Aug;31(4):477-489. doi: 10.1007/s10286-021-00787-5. Epub 2021 Feb 28. Clin Auton Res. 2021. PMID: 33641054 Free PMC article. Review.
-
Expression of MHC II in DRG neurons attenuates paclitaxel-induced cold hypersensitivity in male and female mice.PLoS One. 2024 Feb 8;19(2):e0298396. doi: 10.1371/journal.pone.0298396. eCollection 2024. PLoS One. 2024. PMID: 38330029 Free PMC article.
-
Medical Devices for Tremor Suppression: Current Status and Future Directions.Biosensors (Basel). 2021 Mar 30;11(4):99. doi: 10.3390/bios11040099. Biosensors (Basel). 2021. PMID: 33808056 Free PMC article. Review.
-
Contemporary views on inflammatory pain mechanisms: TRPing over innate and microglial pathways.F1000Res. 2016 Sep 30;5:F1000 Faculty Rev-2425. doi: 10.12688/f1000research.8710.1. eCollection 2016. F1000Res. 2016. PMID: 27781082 Free PMC article. Review.
-
Oxidized Lipids in Persistent Pain States.Front Pharmacol. 2019 Oct 15;10:1147. doi: 10.3389/fphar.2019.01147. eCollection 2019. Front Pharmacol. 2019. PMID: 31680947 Free PMC article. Review.
References
-
- Ossipov MH, Bian D, Malan TP, Jr, Lai J, Porreca F. Lack of involvement of capsaicin-sensitive primary afferents in nerve-ligation injury induced tactile allodynia in rats. Pain. 1999;79:127–133. - PubMed
-
- Liu CN, et al. Tactile allodynia in the absence of C-fiber activation: altered firing properties of DRG neurons following spinal nerve injury. Pain. 2000;85:503–521. - PubMed
-
- Koltzenburg M, Lundberg LE, Torebjork HE. Dynamic and static components of mechanical hyperalgesia in human hairy skin. Pain. 1992;51:207–219. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS087988/NS/NINDS NIH HHS/United States
- R01 NS87988/NS/NINDS NIH HHS/United States
- R01 DE022743/DE/NIDCR NIH HHS/United States
- R01 NS89479/NS/NINDS NIH HHS/United States
- R01 DE19440/DE/NIDCR NIH HHS/United States
- R01 DE019440/DE/NIDCR NIH HHS/United States
- R01 DE017794/DE/NIDCR NIH HHS/United States
- R01 NS067686/NS/NINDS NIH HHS/United States
- R01 NS67686/NS/NINDS NIH HHS/United States
- R21 NS91779/NS/NINDS NIH HHS/United States
- R21 NS082985/NS/NINDS NIH HHS/United States
- R01 NS089479/NS/NINDS NIH HHS/United States
- R01 DE22743/DE/NIDCR NIH HHS/United States
- R21 NS82985/NS/NINDS NIH HHS/United States
- R21 NS091779/NS/NINDS NIH HHS/United States
- R01 DE17794/DE/NIDCR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

