Sirt6 depletion causes spindle defects and chromosome misalignment during meiosis of mouse oocyte
- PMID: 26481302
- PMCID: PMC4612726
- DOI: 10.1038/srep15366
Sirt6 depletion causes spindle defects and chromosome misalignment during meiosis of mouse oocyte
Abstract
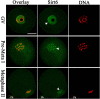
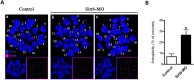
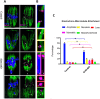
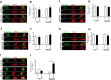
Sirt6, a member of the sirtuin family of NAD-dependent protein deacetylases, has been implicated in multiple biological processes. However, the roles of Sirt6 in meiosis have not been addressed. In the present study, by employing knockdown analysis in mouse oocytes, we evaluated the effects of Sirt6 on meiotic apparatus. We found that specific depletion of Sirt6 results in disruption of spindle morphology and chromosome alignment in oocytes. Consistent with this observation, incidence of aneuploidy is also markedly increased in Sirt6-depleted oocytes. Furthermore, confocal scanning showed that kinetochore-microtubule interaction, an important mechanism controlling chromosome segregation, is severely impaired in metaphase oocytes following Sirt6 knockdown. Unexpectedly, we discovered that Sirt6 modulates the acetylation status of histone H4K16 as their knockdown specifically induces the hyperacetylation of H4K16 in oocytes, which may be associated with the defective phenotypes described above via altering kinetochore function. Altogether, our data reveal a novel function of Sirt6 during oocyte meiosis and indicate a pathway regulating meiotic apparatus.
Figures


Similar articles
-
HDAC3 promotes meiotic apparatus assembly in mouse oocytes by modulating tubulin acetylation.Development. 2017 Oct 15;144(20):3789-3797. doi: 10.1242/dev.153353. Epub 2017 Sep 21. Development. 2017. PMID: 28935703
-
HDAC11 promotes meiotic apparatus assembly during mouse oocyte maturation via decreasing H4K16 and α-tubulin acetylation.Cell Cycle. 2020 Feb;19(3):354-362. doi: 10.1080/15384101.2019.1711315. Epub 2020 Jan 7. Cell Cycle. 2020. PMID: 31910069 Free PMC article.
-
Rab6a is a novel regulator of meiotic apparatus and maturational progression in mouse oocytes.Sci Rep. 2016 Feb 26;6:22209. doi: 10.1038/srep22209. Sci Rep. 2016. PMID: 26915694 Free PMC article.
-
Meiotic defects in human oocytes: Potential causes and clinical implications.Bioessays. 2022 Dec;44(12):e2200135. doi: 10.1002/bies.202200135. Epub 2022 Oct 7. Bioessays. 2022. PMID: 36207289 Review.
-
Aneuploidy in human eggs: contributions of the meiotic spindle.Biochem Soc Trans. 2021 Feb 26;49(1):107-118. doi: 10.1042/BST20200043. Biochem Soc Trans. 2021. PMID: 33449109 Free PMC article. Review.
Cited by
-
SIRT6 Maintains Redox Homeostasis to Promote Porcine Oocyte Maturation.Front Cell Dev Biol. 2021 Feb 25;9:625540. doi: 10.3389/fcell.2021.625540. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33718364 Free PMC article.
-
SIRT6 deficiency causes ovarian hypoplasia by affecting Plod1-related collagen formation.Aging Cell. 2024 Feb;23(2):e14031. doi: 10.1111/acel.14031. Epub 2023 Nov 7. Aging Cell. 2024. PMID: 37936548 Free PMC article.
-
Histone acetyltransferase KAT8 is essential for mouse oocyte development by regulating reactive oxygen species levels.Development. 2017 Jun 15;144(12):2165-2174. doi: 10.1242/dev.149518. Epub 2017 May 15. Development. 2017. PMID: 28506985 Free PMC article.
-
NAD+, Sirtuins and PARPs: enhancing oocyte developmental competence.J Reprod Dev. 2022 Dec 19;68(6):345-354. doi: 10.1262/jrd.2022-052. Epub 2022 Sep 27. J Reprod Dev. 2022. PMID: 36171094 Free PMC article. Review.
-
Insights into the NAD+ biosynthesis pathways involved during meiotic maturation and spindle formation in porcine oocytes.J Reprod Dev. 2022 Jun 1;68(3):216-224. doi: 10.1262/jrd.2021-130. Epub 2022 Mar 28. J Reprod Dev. 2022. PMID: 35342119 Free PMC article.
References
-
- Eppig J. J. Coordination of nuclear and cytoplasmic oocyte maturation in eutherian mammals. Reprod Fertil Dev 8, 485–489 (1996). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

