DMP-1-mediated Ghr gene recombination compromises skeletal development and impairs skeletal response to intermittent PTH
- PMID: 26481310
- PMCID: PMC4714547
- DOI: 10.1096/fj.15-275859
DMP-1-mediated Ghr gene recombination compromises skeletal development and impairs skeletal response to intermittent PTH
Abstract
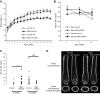
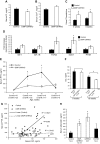
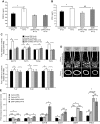
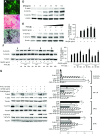
Bone minerals are acquired during growth and are key determinants of adult skeletal health. During puberty, the serum levels of growth hormone (GH) and its downstream effector IGF-1 increase and play critical roles in bone acquisition. The goal of the current study was to determine how bone cells integrate signals from the GH/IGF-1 to enhance skeletal mineralization and strength during pubertal growth. Osteocytes, the most abundant bone cells, were shown to orchestrate bone modeling during growth. We used dentin matrix protein (Dmp)-1-mediated Ghr knockout (DMP-GHRKO) mice to address the role of the GH/IGF axis in osteocytes. We found that DMP-GHRKO did not affect linear growth but compromised overall bone accrual. DMP-GHRKO mice exhibited reduced serum inorganic phosphate and parathyroid hormone (PTH) levels and decreased bone formation indices and were associated with an impaired response to intermittent PTH treatment. Using an osteocyte-like cell line along with in vivo studies, we found that PTH sensitized the response of bone to GH by increasing Janus kinase-2 and IGF-1R protein levels. We concluded that endogenously secreted PTH and GHR signaling in bone are necessary to establish radial bone growth and optimize mineral acquisition during growth.
Keywords: fibroblast growth factor-23; growth hormone receptor; microcomputed tomography; osteocyte; parathyroid hormone.
© FASEB.
Figures

Similar articles
-
Growth without growth hormone receptor: estradiol is a major growth hormone-independent regulator of hepatic IGF-I synthesis.J Bone Miner Res. 2005 Dec;20(12):2138-49. doi: 10.1359/JBMR.050811. Epub 2005 Aug 8. J Bone Miner Res. 2005. PMID: 16294267
-
Impact of androgens, growth hormone, and IGF-I on bone and muscle in male mice during puberty.J Bone Miner Res. 2007 Jan;22(1):72-82. doi: 10.1359/jbmr.060911. J Bone Miner Res. 2007. PMID: 17014385
-
Does the GH/IGF-1 axis contribute to skeletal sexual dimorphism? Evidence from mouse studies.Growth Horm IGF Res. 2016 Apr;27:7-17. doi: 10.1016/j.ghir.2015.12.004. Epub 2015 Dec 31. Growth Horm IGF Res. 2016. PMID: 26843472 Free PMC article. Review.
-
The ternary IGF complex influences postnatal bone acquisition and the skeletal response to intermittent parathyroid hormone.J Endocrinol. 2006 May;189(2):289-99. doi: 10.1677/joe.1.06657. J Endocrinol. 2006. PMID: 16648296
-
Bone mineralization is regulated by signaling cross talk between molecular factors of local and systemic origin: the role of fibroblast growth factor 23.Biofactors. 2014 Nov-Dec;40(6):555-68. doi: 10.1002/biof.1186. Epub 2014 Oct 29. Biofactors. 2014. PMID: 25352227 Review.
Cited by
-
Tissue-Specific GHR Knockout Mice: An Updated Review.Front Endocrinol (Lausanne). 2020 Oct 9;11:579909. doi: 10.3389/fendo.2020.579909. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 33162937 Free PMC article. Review.
-
Mitochondrial Function Is Compromised in Cortical Bone Osteocytes of Long-Lived Growth Hormone Receptor Null Mice.J Bone Miner Res. 2019 Jan;34(1):106-122. doi: 10.1002/jbmr.3573. Epub 2018 Sep 14. J Bone Miner Res. 2019. PMID: 30216544 Free PMC article.
-
Mouse models of growth hormone insensitivity.Rev Endocr Metab Disord. 2021 Mar;22(1):17-29. doi: 10.1007/s11154-020-09600-6. Epub 2020 Oct 10. Rev Endocr Metab Disord. 2021. PMID: 33037595 Free PMC article. Review.
-
Advanced Skeletal Ossification Is Associated with Genetic Variants in Chronologically Young Beef Heifers.Genes (Basel). 2023 Aug 15;14(8):1629. doi: 10.3390/genes14081629. Genes (Basel). 2023. PMID: 37628680 Free PMC article.
-
Physiological Bone Remodeling: Systemic Regulation and Growth Factor Involvement.Physiology (Bethesda). 2016 May;31(3):233-45. doi: 10.1152/physiol.00061.2014. Physiology (Bethesda). 2016. PMID: 27053737 Free PMC article. Review.
References
-
- Harvey N., Dennison E., Cooper C. (2010) Osteoporosis: impact on health and economics. Nat. Rev. Rheumatol. 6, 99–105 - PubMed
-
- Erlandson M. C., Kontulainen S. A., Chilibeck P. D., Arnold C. M., Faulkner R. A., Baxter-Jones A. D. (2012) Former premenarcheal gymnasts exhibit site-specific skeletal benefits in adulthood after long-term retirement. J. Bone Miner. Res. 27, 2298–2305 - PubMed
-
- Ferrari S., Rizzoli R., Slosman D., Bonjour J. P. (1998) Familial resemblance for bone mineral mass is expressed before puberty. J. Clin. Endocrinol. Metab. 83, 358–361 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

