Neuroimaging of Natalizumab Complications in Multiple Sclerosis: PML and Other Associated Entities
- PMID: 26483978
- PMCID: PMC4592919
- DOI: 10.1155/2015/809252
Neuroimaging of Natalizumab Complications in Multiple Sclerosis: PML and Other Associated Entities
Abstract
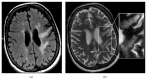
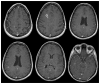
Natalizumab (Tysabri) is a monoclonal antibody (α4 integrin antagonist) approved for treatment of multiple sclerosis, both for patients who fail therapy with other disease modifying agents and for patients with aggressive disease. Natalizumab is highly effective, resulting in significant decreases in rates of both relapse and disability accumulation, as well as marked decrease in MRI evidence of disease activity. As such, utilization of natalizumab is increasing, and the presentation of its associated complications is increasing accordingly. This review focuses on the clinical and neuroimaging features of the major complications associated with natalizumab therapy, focusing on the rare but devastating progressive multifocal leukoencephalopathy (PML). Associated entities including PML associated immune reconstitution inflammatory syndrome (PML-IRIS) and the emerging phenomenon of rebound of MS disease activity after natalizumab discontinuation are also discussed. Early recognition of neuroimaging features associated with these processes is critical in order to facilitate prompt diagnosis, treatment, and/or modification of therapies to improve patient outcomes.
Figures








References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

