Stabilizing versus destabilizing the microtubules: a double-edge sword for an effective cancer treatment option?
- PMID: 26484003
- PMCID: PMC4592889
- DOI: 10.1155/2015/690916
Stabilizing versus destabilizing the microtubules: a double-edge sword for an effective cancer treatment option?
Abstract
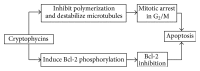
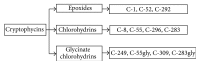
Microtubules are dynamic and structural cellular components involved in several cell functions, including cell shape, motility, and intracellular trafficking. In proliferating cells, they are essential components in the division process through the formation of the mitotic spindle. As a result of these functions, tubulin and microtubules are targets for anticancer agents. Microtubule-targeting agents can be divided into two groups: microtubule-stabilizing, and microtubule-destabilizing agents. The former bind to the tubulin polymer and stabilize microtubules, while the latter bind to the tubulin dimers and destabilize microtubules. Alteration of tubulin-microtubule equilibrium determines the disruption of the mitotic spindle, halting the cell cycle at the metaphase-anaphase transition and, eventually, resulting in cell death. Clinical application of earlier microtubule inhibitors, however, unfortunately showed several limits, such as neurological and bone marrow toxicity and the emergence of drug-resistant tumor cells. Here we review several natural and synthetic microtubule-targeting agents, which showed antitumor activity and increased efficacy in comparison to traditional drugs in various preclinical and clinical studies. Cryptophycins, combretastatins, ombrabulin, soblidotin, D-24851, epothilones and discodermolide were used in clinical trials. Some of them showed antiangiogenic and antivascular activity and others showed the ability to overcome multidrug resistance, supporting their possible use in chemotherapy.
Figures


Similar articles
-
Microtubule Targeting Agents as Cancer Chemotherapeutics: An Overview of Molecular Hybrids as Stabilizing and Destabilizing Agents.Curr Top Med Chem. 2017;17(22):2523-2537. doi: 10.2174/1568026617666170104145640. Curr Top Med Chem. 2017. PMID: 28056738 Review.
-
Mechanism of action of antitumor drugs that interact with microtubules and tubulin.Curr Med Chem Anticancer Agents. 2002 Jan;2(1):1-17. doi: 10.2174/1568011023354290. Curr Med Chem Anticancer Agents. 2002. PMID: 12678749 Review.
-
Vinflunine: a new vision that may translate into antiangiogenic and antimetastatic activity.Anticancer Drugs. 2012 Jan;23(1):1-11. doi: 10.1097/CAD.0b013e32834d237b. Anticancer Drugs. 2012. PMID: 22027536 Review.
-
Recent advances in microtubule-stabilizing agents.Eur J Med Chem. 2018 Jan 1;143:806-828. doi: 10.1016/j.ejmech.2017.11.062. Epub 2017 Nov 24. Eur J Med Chem. 2018. PMID: 29223097 Review.
-
Microtubule inhibitors: Differentiating tubulin-inhibiting agents based on mechanisms of action, clinical activity, and resistance.Mol Cancer Ther. 2009 Aug;8(8):2086-95. doi: 10.1158/1535-7163.MCT-09-0366. Epub 2009 Aug 11. Mol Cancer Ther. 2009. PMID: 19671735 Review.
Cited by
-
Depletion of JMJD5 sensitizes tumor cells to microtubule-destabilizing agents by altering microtubule stability.Cell Cycle. 2016 Nov;15(21):2980-2991. doi: 10.1080/15384101.2016.1234548. Epub 2016 Oct 7. Cell Cycle. 2016. PMID: 27715397 Free PMC article.
-
Depsipeptides Targeting Tumor Cells: Milestones from In Vitro to Clinical Trials.Molecules. 2023 Jan 9;28(2):670. doi: 10.3390/molecules28020670. Molecules. 2023. PMID: 36677728 Free PMC article. Review.
-
Reorganization of paclitaxel-stabilized microtubule arrays at mitotic entry: roles of depolymerizing kinesins and severing proteins.Cancer Biol Ther. 2019;20(10):1337-1347. doi: 10.1080/15384047.2019.1638678. Epub 2019 Jul 25. Cancer Biol Ther. 2019. PMID: 31345098 Free PMC article.
-
Human MAP Tau Based Targeted Cytolytic Fusion Proteins.Biomedicines. 2017 Jun 27;5(3):36. doi: 10.3390/biomedicines5030036. Biomedicines. 2017. PMID: 28653985 Free PMC article. Review.
-
Thermosensitive Betulinic Acid-Loaded Magnetoliposomes: A Promising Antitumor Potential for Highly Aggressive Human Breast Adenocarcinoma Cells Under Hyperthermic Conditions.Int J Nanomedicine. 2020 Oct 23;15:8175-8200. doi: 10.2147/IJN.S269630. eCollection 2020. Int J Nanomedicine. 2020. PMID: 33122905 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

