Loss of Glycosaminoglycan Receptor Binding after Mosquito Cell Passage Reduces Chikungunya Virus Infectivity
- PMID: 26484530
- PMCID: PMC4615622
- DOI: 10.1371/journal.pntd.0004139
Loss of Glycosaminoglycan Receptor Binding after Mosquito Cell Passage Reduces Chikungunya Virus Infectivity
Abstract
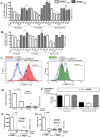
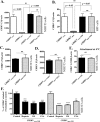
Chikungunya virus (CHIKV) is a mosquito-transmitted alphavirus that can cause fever and chronic arthritis in humans. CHIKV that is generated in mosquito or mammalian cells differs in glycosylation patterns of viral proteins, which may affect its replication and virulence. Herein, we compare replication, pathogenicity, and receptor binding of CHIKV generated in Vero cells (mammal) or C6/36 cells (mosquito) through a single passage. We demonstrate that mosquito cell-derived CHIKV (CHIKV mos) has slower replication than mammalian cell-derived CHIKV (CHIKV vero), when tested in both human and murine cell lines. Consistent with this, CHIKV mos infection in both cell lines produce less cytopathic effects and reduced antiviral responses. In addition, infection in mice show that CHIKV mos produces a lower level of viremia and less severe footpad swelling when compared with CHIKV vero. Interestingly, CHIKV mos has impaired ability to bind to glycosaminoglycan (GAG) receptors on mammalian cells. However, sequencing analysis shows that this impairment is not due to a mutation in the CHIKV E2 gene, which encodes for the viral receptor binding protein. Moreover, CHIKV mos progenies can regain GAG receptor binding capability and can replicate similarly to CHIKV vero after a single passage in mammalian cells. Furthermore, CHIKV vero and CHIKV mos no longer differ in replication when N-glycosylation of viral proteins was inhibited by growing these viruses in the presence of tunicamycin. Collectively, these results suggest that N-glycosylation of viral proteins within mosquito cells can result in loss of GAG receptor binding capability of CHIKV and reduction of its infectivity in mammalian cells.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Gauri LA, Ranwa BL, Nagar K, Vyas A, Fatima Q (2012) Post chikungunya brain stem encephalitis. J Assoc Physicians India 60: 68–70. - PubMed
-
- Pellot AS, Alessandri JL, Robin S, Samperiz S, Attali T, et al. (2012) [Severe forms of chikungunya virus infection in a pediatric intensive care unit on Reunion Island]. Med Trop (Mars) 72 Spec No: 88–93. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

