Canonical Poly(A) Polymerase Activity Promotes the Decay of a Wide Variety of Mammalian Nuclear RNAs
- PMID: 26484760
- PMCID: PMC4618350
- DOI: 10.1371/journal.pgen.1005610
Canonical Poly(A) Polymerase Activity Promotes the Decay of a Wide Variety of Mammalian Nuclear RNAs
Abstract
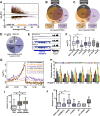
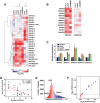
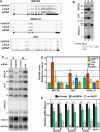
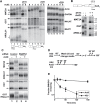
The human nuclear poly(A)-binding protein PABPN1 has been implicated in the decay of nuclear noncoding RNAs (ncRNAs). In addition, PABPN1 promotes hyperadenylation by stimulating poly(A)-polymerases (PAPα/γ), but this activity has not previously been linked to the decay of endogenous transcripts. Moreover, the mechanisms underlying target specificity have remained elusive. Here, we inactivated PAP-dependent hyperadenylation in cells by two independent mechanisms and used an RNA-seq approach to identify endogenous targets. We observed the upregulation of various ncRNAs, including snoRNA host genes, primary miRNA transcripts, and promoter upstream antisense RNAs, confirming that hyperadenylation is broadly required for the degradation of PABPN1-targets. In addition, we found that mRNAs with retained introns are susceptible to PABPN1 and PAPα/γ-mediated decay (PPD). Transcripts are targeted for degradation due to inefficient export, which is a consequence of reduced intron number or incomplete splicing. Additional investigation showed that a genetically-encoded poly(A) tail is sufficient to drive decay, suggesting that degradation occurs independently of the canonical cleavage and polyadenylation reaction. Surprisingly, treatment with transcription inhibitors uncouples polyadenylation from decay, leading to runaway hyperadenylation of nuclear decay targets. We conclude that PPD is an important mammalian nuclear RNA decay pathway for the removal of poorly spliced and nuclear-retained transcripts.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
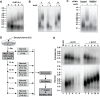
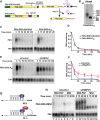
Similar articles
-
The human nuclear poly(a)-binding protein promotes RNA hyperadenylation and decay.PLoS Genet. 2013;9(10):e1003893. doi: 10.1371/journal.pgen.1003893. Epub 2013 Oct 17. PLoS Genet. 2013. PMID: 24146636 Free PMC article.
-
PABPN1 prevents the nuclear export of an unspliced RNA with a constitutive transport element and controls human gene expression via intron retention.RNA. 2023 May;29(5):644-662. doi: 10.1261/rna.079294.122. Epub 2023 Feb 8. RNA. 2023. PMID: 36754576 Free PMC article.
-
Polyadenylation-dependent control of long noncoding RNA expression by the poly(A)-binding protein nuclear 1.PLoS Genet. 2012;8(11):e1003078. doi: 10.1371/journal.pgen.1003078. Epub 2012 Nov 15. PLoS Genet. 2012. PMID: 23166521 Free PMC article.
-
Implications of polyadenylation in health and disease.Nucleus. 2014;5(6):508-19. doi: 10.4161/nucl.36360. Epub 2014 Oct 31. Nucleus. 2014. PMID: 25484187 Free PMC article. Review.
-
Long noncoding RNAs: Re-writing dogmas of RNA processing and stability.Biochim Biophys Acta. 2016 Jan;1859(1):128-38. doi: 10.1016/j.bbagrm.2015.06.003. Epub 2015 Jun 11. Biochim Biophys Acta. 2016. PMID: 26073320 Free PMC article. Review.
Cited by
-
The Role of the m6A RNA Methyltransferase METTL16 in Gene Expression and SAM Homeostasis.Genes (Basel). 2022 Dec 8;13(12):2312. doi: 10.3390/genes13122312. Genes (Basel). 2022. PMID: 36553579 Free PMC article. Review.
-
O-GlcNAc regulates gene expression by controlling detained intron splicing.Nucleic Acids Res. 2020 Jun 4;48(10):5656-5669. doi: 10.1093/nar/gkaa263. Nucleic Acids Res. 2020. PMID: 32329777 Free PMC article.
-
hnRNPH1-MTR4 complex-mediated regulation of NEAT1v2 stability is critical for IL8 expression.RNA Biol. 2021 Oct 15;18(sup1):537-547. doi: 10.1080/15476286.2021.1971439. Epub 2021 Sep 1. RNA Biol. 2021. PMID: 34470577 Free PMC article.
-
Genetic regulation of nascent RNA maturation revealed by direct RNA nanopore sequencing.bioRxiv [Preprint]. 2024 Aug 29:2024.08.29.610338. doi: 10.1101/2024.08.29.610338. bioRxiv. 2024. Update in: Genome Res. 2025 Apr 14;35(4):712-724. doi: 10.1101/gr.279203.124. PMID: 39257732 Free PMC article. Updated. Preprint.
-
Genetic regulation of nascent RNA maturation revealed by direct RNA nanopore sequencing.Genome Res. 2025 Apr 14;35(4):712-724. doi: 10.1101/gr.279203.124. Genome Res. 2025. PMID: 39952678
References
-
- Fasken MB, Corbett AH. Mechanisms of nuclear mRNA quality control. RNA Biol. 2009;6: 237–241. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

