Singlet Oxygen-Mediated Oxidation during UVA Radiation Alters the Dynamic of Genomic DNA Replication
- PMID: 26485711
- PMCID: PMC4618472
- DOI: 10.1371/journal.pone.0140645
Singlet Oxygen-Mediated Oxidation during UVA Radiation Alters the Dynamic of Genomic DNA Replication
Abstract
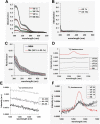
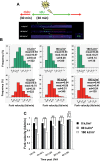
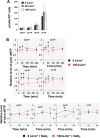
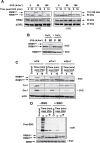
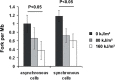
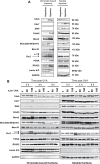
UVA radiation (320-400 nm) is a major environmental agent that can exert its deleterious action on living organisms through absorption of the UVA photons by endogenous or exogenous photosensitizers. This leads to the production of reactive oxygen species (ROS), such as singlet oxygen (1O2) and hydrogen peroxide (H2O2), which in turn can modify reversibly or irreversibly biomolecules, such as lipids, proteins and nucleic acids. We have previously reported that UVA-induced ROS strongly inhibit DNA replication in a dose-dependent manner, but independently of the cell cycle checkpoints activation. Here, we report that the production of 1O2 by UVA radiation leads to a transient inhibition of replication fork velocity, a transient decrease in the dNTP pool, a quickly reversible GSH-dependent oxidation of the RRM1 subunit of ribonucleotide reductase and sustained inhibition of origin firing. The time of recovery post irradiation for each of these events can last from few minutes (reduction of oxidized RRM1) to several hours (replication fork velocity and origin firing). The quenching of 1O2 by sodium azide prevents the delay of DNA replication, the decrease in the dNTP pool and the oxidation of RRM1, while inhibition of Chk1 does not prevent the inhibition of origin firing. Although the molecular mechanism remains elusive, our data demonstrate that the dynamic of replication is altered by UVA photosensitization of vitamins via the production of singlet oxygen.
Conflict of interest statement
Figures

Similar articles
-
Hydrogen peroxide is responsible for UVA-induced DNA damage measured by alkaline comet assay in HaCaT keratinocytes.J Photochem Photobiol B. 2000 Dec;59(1-3):123-31. doi: 10.1016/s1011-1344(00)00149-4. J Photochem Photobiol B. 2000. PMID: 11332879
-
Inhibition of S-phase progression triggered by UVA-induced ROS does not require a functional DNA damage checkpoint response in mammalian cells.DNA Repair (Amst). 2008 Sep 1;7(9):1500-16. doi: 10.1016/j.dnarep.2008.05.004. Epub 2008 Jul 7. DNA Repair (Amst). 2008. PMID: 18603484
-
UVA irradiation of fatty acids and their oxidized products substantially increases their ability to generate singlet oxygen.Phys Chem Chem Phys. 2013 Oct 28;15(40):17672-80. doi: 10.1039/c3cp51399h. Phys Chem Chem Phys. 2013. PMID: 24043219
-
Photosensitized DNA damage and its protection via a novel mechanism.Photochem Photobiol. 2007 Jan-Feb;83(1):205-12. doi: 10.1562/2006-03-09-IR-840. Photochem Photobiol. 2007. PMID: 16965181 Review.
-
UVA and endogenous photosensitizers--the detection of singlet oxygen by its luminescence.Photochem Photobiol Sci. 2012 Jan;11(1):107-17. doi: 10.1039/c1pp05142c. Epub 2011 Oct 11. Photochem Photobiol Sci. 2012. PMID: 21986813 Review.
Cited by
-
Long-wavelength UVA enhances UVB-induced cell death in cultured keratinocytes: DSB formation and suppressed survival pathway.Photochem Photobiol Sci. 2021 May;20(5):639-652. doi: 10.1007/s43630-021-00050-w. Epub 2021 May 12. Photochem Photobiol Sci. 2021. PMID: 33978941
-
Redox modifications of cysteine-containing proteins, cell cycle arrest and translation inhibition: Involvement in vitamin C-induced breast cancer cell death.Redox Biol. 2019 Sep;26:101290. doi: 10.1016/j.redox.2019.101290. Epub 2019 Aug 2. Redox Biol. 2019. PMID: 31412312 Free PMC article.
-
Differential correlations between changes to glutathione redox state, protein ubiquitination, and stress-inducible HSPA chaperone expression after different types of oxidative stress.Cell Stress Chaperones. 2018 Sep;23(5):985-1002. doi: 10.1007/s12192-018-0909-y. Epub 2018 May 12. Cell Stress Chaperones. 2018. PMID: 29754332 Free PMC article.
-
Direct 1O2 optical excitation: A tool for redox biology.Redox Biol. 2017 Oct;13:39-59. doi: 10.1016/j.redox.2017.05.011. Epub 2017 May 25. Redox Biol. 2017. PMID: 28570948 Free PMC article. Review.
-
XRRA1 Targets ATM/CHK1/2-Mediated DNA Repair in Colorectal Cancer.Biomed Res Int. 2017;2017:5718968. doi: 10.1155/2017/5718968. Epub 2017 Sep 26. Biomed Res Int. 2017. PMID: 29082250 Free PMC article.
References
-
- Madronich S, McKenzie RL, Bjorn LO, Caldwell MM (1998) Changes in biologically active ultraviolet radiation reaching the Earth's surface. J Photochem Photobiol B 46: 5–19. - PubMed
-
- Wondrak GT, Jacobson MK, Jacobson EL (2006) Endogenous UVA-photosensitizers: mediators of skin photodamage and novel targets for skin photoprotection. Photochem Photobiol Sci 5: 215–237. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

