Postural responses to anterior and posterior perturbations applied to the upper trunk of standing human subjects
- PMID: 26487178
- PMCID: PMC4731437
- DOI: 10.1007/s00221-015-4442-2
Postural responses to anterior and posterior perturbations applied to the upper trunk of standing human subjects
Abstract
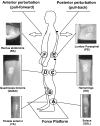
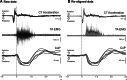
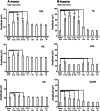
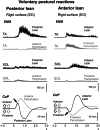
This study concerned the effects of brisk perturbations applied to the shoulders of standing subjects to displace them either forwards or backwards, our aim being to characterise the responses to these disturbances. Subjects stood on a force platform, and acceleration was measured at the level of C7, the sacrum and both tibial tuberosities. Surface EMG was measured from soleus (SOL), tibialis anterior (TA), the hamstrings (HS), quadriceps (QUAD), rectus abdominis (RA) and lumbar paraspinal (PS) muscles. Trials were recorded for each of four conditions: subjects' eyes open (reference) or closed and on a firm (reference) or compliant surface. Observations were also made of voluntary postural reactions to a tap over the deltoid. Anterior perturbations (mean C7 acceleration 251.7 mg) evoked activity within the dorsal muscles (SOL, HS, PS) with a similar latency to voluntary responses to shoulder tapping. Responses to posterior perturbations (mean C7 acceleration -240.4 mg) were more complex beginning, on average, at shorter latency than voluntary activity (median TA 78.0 ms). There was activation of TA, QUAD and SOL associated with initial forward acceleration of the lower legs. The EMG responses consisted of an initial phasic discharge followed by a more prolonged one. These responses differ from the pattern of automatic postural responses that follow displacements at the level of the ankles, and it is unlikely that proprioceptive afferents excited by ankle movement had a role in the initial responses. Vision and surface properties had only minor effects. Perturbations of the upper trunk evoke stereotyped compensatory postural responses for each direction of perturbation. For posterior perturbations, EMG onset occurs earlier than for voluntary responses.
Figures

Similar articles
-
Triggering of balance corrections and compensatory strategies in a patient with total leg proprioceptive loss.Exp Brain Res. 2002 Jan;142(1):91-107. doi: 10.1007/s00221-001-0926-3. Epub 2001 Nov 14. Exp Brain Res. 2002. PMID: 11797087
-
Responses to anterior and posterior perturbations in Parkinson's disease with early postural instability: role of axial and limb rigidity.Exp Brain Res. 2019 Jul;237(7):1853-1867. doi: 10.1007/s00221-019-05553-8. Epub 2019 May 13. Exp Brain Res. 2019. PMID: 31087110
-
Vestibular influences on human postural control in combinations of pitch and roll planes reveal differences in spatiotemporal processing.Exp Brain Res. 2001 Sep;140(1):95-111. doi: 10.1007/s002210100802. Exp Brain Res. 2001. PMID: 11500802
-
Is lower leg proprioception essential for triggering human automatic postural responses?Exp Brain Res. 2000 Feb;130(3):375-91. doi: 10.1007/s002219900259. Exp Brain Res. 2000. PMID: 10706436
-
Cortical control of postural responses.J Neural Transm (Vienna). 2007;114(10):1339-48. doi: 10.1007/s00702-007-0657-0. Epub 2007 Mar 29. J Neural Transm (Vienna). 2007. PMID: 17393068 Free PMC article. Review.
Cited by
-
Neurophysiological analysis of the clinical pull test.J Neurophysiol. 2018 Nov 1;120(5):2325-2333. doi: 10.1152/jn.00789.2017. Epub 2018 Aug 15. J Neurophysiol. 2018. PMID: 30110235 Free PMC article.
-
Wobble Board Instability Enhances Compensatory CoP Responses to CoM Movement Across Timescales.Sensors (Basel). 2025 Jul 17;25(14):4454. doi: 10.3390/s25144454. Sensors (Basel). 2025. PMID: 40732579 Free PMC article.
-
Age-Related Differences in Stepping Reactions to a Balance Perturbation: A Functional Near-Infrared Spectroscopy and Surface Electromyography Study.Brain Sci. 2022 Oct 31;12(11):1479. doi: 10.3390/brainsci12111479. Brain Sci. 2022. PMID: 36358405 Free PMC article.
-
Center of pressure displacement due to graded controlled perturbations to the trunk in standing subjects: the force-impulse paradigm.Eur J Appl Physiol. 2022 Feb;122(2):425-435. doi: 10.1007/s00421-021-04844-9. Epub 2021 Nov 19. Eur J Appl Physiol. 2022. PMID: 34797437
-
Response to perturbation during quiet standing resembles delayed state feedback optimized for performance and robustness.Sci Rep. 2021 May 31;11(1):11392. doi: 10.1038/s41598-021-90305-4. Sci Rep. 2021. PMID: 34059718 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

