Electrical stimulation therapy of the lower esophageal sphincter is successful in treating GERD: long-term 3-year results
- PMID: 26487200
- PMCID: PMC4912595
- DOI: 10.1007/s00464-015-4539-5
Electrical stimulation therapy of the lower esophageal sphincter is successful in treating GERD: long-term 3-year results
Abstract
Background: Electrical stimulation of the lower esophageal sphincter (LES) has been shown to improve outcomes in patients with gastroesophageal reflux disease (GERD) at 2 years. The aim of the study was to evaluate the safety and efficacy of LES stimulation in the same cohort at 3 years.
Methods: GERD patients with partial response to PPI, with % 24-h esophageal pH < 4.0 for >5 %, with hiatal hernia <3 cm and with esophagitis ≤LA grade C were treated with LES stimulation in an open-label 2-year trial. All patients were on fixed stimulation parameter of 20 Hz, 220 μs, 5 mA delivered in twelve, 30-min sessions. After completing the 2-year open-label study, they were offered enrollment into a multicenter registry trial and were evaluated using GERD-HRQL, symptom diaries and pH testing at their 3-year follow-up.
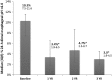
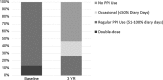
Results: Fifteen patients completed their 3-year evaluation [mean (SD) age = 56.1 (9.7) years; men = 8] on LES stimulation. At 3 years, there was a significant improvement in their median (IQR) GERD-HRQL on electrical stimulation compared to both their on PPI [9 (6-10) vs. 1 (0-2), p = 0.001] and off PPI [22 (21-24) vs. 1 (0-2), p < 0.001]. Median 24-h distal esophageal acid exposure was significantly reduced from [10.3 (7.5-11.6) % at baseline vs. 3 (1.9-4.5) %, p < 0.001] at 3 years. Seventy-three % (11/15) patients had normalized their distal esophageal acid exposure at 3 years. Remaining four patients had improved their distal esophageal acid exposure by 39-48 % from baseline. All but four patients reported cessation of regular PPI use (>50 % of days with PPI use); three had normal esophageal pH at 3 years. There were no unanticipated device- or stimulation-related adverse events or untoward sensation reported during the 2- to 3-year follow-up.
Conclusion: LES-EST is safe and effective for treating patients with GERD over long-term, 3-year duration. There was a significant and sustained improvement in esophageal acid exposure and reduction in GERD symptoms and PPI use. Further, no new GI side effects or adverse events were reported.
Keywords: Electrical stimulation; Esophageal acid; GERD; Lower esophageal sphincter; Outcomes; Treatment.
Figures




References
-
- Jamieson JR, Stein HJ, DeMeester TR, Bonavina L, Schwizer W, Hinder RA, Albertucci M. Ambulatory 24-h esophageal pH monitoring: normal values, optimal thresholds, specificity, sensitivity, and reproducibility. Am J Gastroenterol. 1992;87:1102–1111. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

