Motor Skill Learning Is Associated with Phase-Dependent Modifications in the Striatal cAMP/PKA/DARPP-32 Signaling Pathway in Rodents
- PMID: 26488498
- PMCID: PMC4619563
- DOI: 10.1371/journal.pone.0140974
Motor Skill Learning Is Associated with Phase-Dependent Modifications in the Striatal cAMP/PKA/DARPP-32 Signaling Pathway in Rodents
Abstract
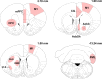
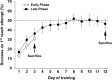
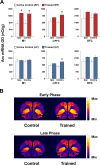
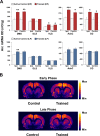
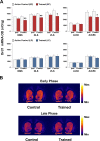
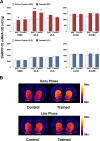
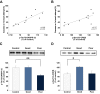
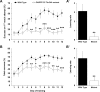
Abundant evidence points to a key role of dopamine in motor skill learning, although the underlying cellular and molecular mechanisms are still poorly understood. Here, we used a skilled-reaching paradigm to first examine changes in the expression of the plasticity-related gene Arc to map activity in cortico-striatal circuitry during different phases of motor skill learning in young animals. In the early phase, Arc mRNA was significantly induced in the medial prefrontal cortex (mPFC), cingulate cortex, primary motor cortex, and striatum. In the late phase, expression of Arc did not change in most regions, except in the mPFC and dorsal striatum. In the second series of experiments, we studied the learning-induced changes in the phosphorylation state of dopamine and cAMP-regulated phosphoprotein, 32k Da (DARPP-32). Western blot analysis of the phosphorylation state of DARPP-32 and its downstream target cAMP response element-binding protein (CREB) in the striatum revealed that the early, but not late, phase of motor skill learning was associated with increased levels of phospho-Thr34-DARPP-32 and phospho-Ser133-CREB. Finally, we used the DARPP-32 knock-in mice with a point mutation in the Thr34 regulatory site (i.e., protein kinase A site) to test the significance of this pathway in motor skill learning. In accordance with our hypothesis, inhibition of DARPP-32 activity at the Thr34 regulatory site strongly attenuated the motor learning rate and skilled reaching performance of mice. These findings suggest that the cAMP/PKA/DARPP-32 signaling pathway is critically involved in the acquisition of novel motor skills, and also demonstrate a dynamic shift in the contribution of cortico-striatal circuitry during different phases of motor skill learning.
Conflict of interest statement
Figures

Similar articles
-
Dopamine D3 receptor deletion increases tissue plasminogen activator (tPA) activity in prefrontal cortex and hippocampus.Neuroscience. 2013 Oct 10;250:546-56. doi: 10.1016/j.neuroscience.2013.07.053. Epub 2013 Jul 29. Neuroscience. 2013. PMID: 23906635
-
Wnt-5a-induced phosphorylation of DARPP-32 inhibits breast cancer cell migration in a CREB-dependent manner.J Biol Chem. 2009 Oct 2;284(40):27533-43. doi: 10.1074/jbc.M109.048884. Epub 2009 Aug 3. J Biol Chem. 2009. PMID: 19651774 Free PMC article.
-
PKA-mediated responses in females' estrous cycle affect cocaine-induced responses in dopamine-mediated intracellular cascades.Neuroscience. 2009 Jul 7;161(3):865-76. doi: 10.1016/j.neuroscience.2009.03.071. Epub 2009 Apr 5. Neuroscience. 2009. PMID: 19348873
-
DARPP-32 and modulation of cAMP signaling: involvement in motor control and levodopa-induced dyskinesia.Parkinsonism Relat Disord. 2004 Jul;10(5):281-6. doi: 10.1016/j.parkreldis.2004.02.010. Parkinsonism Relat Disord. 2004. PMID: 15196506 Review.
-
Psychoactive drugs and regulation of the cAMP/PKA/DARPP-32 cascade in striatal medium spiny neurons.Neurosci Biobehav Rev. 2007;31(1):79-88. doi: 10.1016/j.neubiorev.2006.03.003. Epub 2006 May 30. Neurosci Biobehav Rev. 2007. PMID: 16730373 Review.
Cited by
-
Sucrose Abstinence and Environmental Enrichment Effects on Mesocorticolimbic DARPP32 in Rats.Sci Rep. 2018 Sep 4;8(1):13174. doi: 10.1038/s41598-018-29625-x. Sci Rep. 2018. PMID: 30181585 Free PMC article.
-
Adaptive behaviors of Drosophila larvae on slippery surfaces.J Biol Phys. 2023 Mar;49(1):121-132. doi: 10.1007/s10867-023-09626-2. Epub 2023 Feb 15. J Biol Phys. 2023. PMID: 36790728 Free PMC article.
-
Can Neonatal Systemic Inflammation and Hypoxia Yield a Cerebral Palsy-Like Phenotype in Periadolescent Mice?Mol Neurobiol. 2019 Oct;56(10):6883-6900. doi: 10.1007/s12035-019-1548-8. Epub 2019 Apr 2. Mol Neurobiol. 2019. PMID: 30941732 Free PMC article.
-
Gαo is a major determinant of cAMP signaling in the pathophysiology of movement disorders.Cell Rep. 2021 Feb 2;34(5):108718. doi: 10.1016/j.celrep.2021.108718. Cell Rep. 2021. PMID: 33535037 Free PMC article.
-
Dopamine induces soluble α-synuclein oligomers and nigrostriatal degeneration.Nat Neurosci. 2017 Nov;20(11):1560-1568. doi: 10.1038/nn.4641. Epub 2017 Sep 18. Nat Neurosci. 2017. PMID: 28920936 Free PMC article.
References
-
- Bonnier B, Eliasson AC, Krumlinde-Sundholm L. Effects of constraint-induced movement therapy in adolescents with hemiplegic cerebral palsy: a day camp model. Scand J Occup Ther. 2006;13(1):13–22. . - PubMed
-
- Kleim JA, Barbay S, Nudo RJ. Functional reorganization of the rat motor cortex following motor skill learning. Journal of neurophysiology. 1998;80(6):3321–5. . - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

