Biodegradation and cytotoxicity of ciprofloxacin-loaded hydroxyapatite-polycaprolactone nanocomposite film for sustainable bone implants
- PMID: 26491313
- PMCID: PMC4599610
- DOI: 10.2147/IJN.S79995
Biodegradation and cytotoxicity of ciprofloxacin-loaded hydroxyapatite-polycaprolactone nanocomposite film for sustainable bone implants
Abstract
Introduction: In recent years there has been a steep increase in the number of orthopedic patients for many reasons. One major reason is osteomyelitis, caused by pyrogenic bacteria, with progressive infection of the bone or bone marrow and surrounding tissues. So antibiotics must be introduced during bone implantation to avoid prolonged infection.
Aim: The objective of the study reported here was to prepare a composite film of nanocrystalline hydroxyapatite (HAp) and polycaprolactone (PCL) polymer loaded with ciprofloxacin, a frequently used antibiotic agent for bone infections.
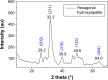
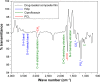
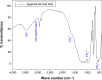
Methods: Nanocrystalline HAp was synthesized by precipitation method using the precursor obtained from eggshell. The nanocomposite film (HAp-PCL-ciprofloxacin) was prepared by solvent evaporation. Drug-release and biodegradation studies were undertaken by immersing the composite film in phosphate-buffered saline solution, while a cytotoxicity test was performed using the fibroblast cell line NIH-3T3 and osteoblast cell line MG-63.
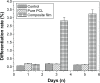
Results: The pure PCL film had quite a low dissolution rate after an initial sharp weight loss, whereas the ciprofloxacin-loaded HAp-PCL nanocomposite film had a large weight loss due to its fast drug release. The composite film had higher water absorption than the pure PCL, and increasing the concentration of the HAp increased the water absorption. The in vitro cell-line study showed a good biocompatibility and bioactivity of the developed nanocomposite film.
Conclusion: The prepared film will act as a sustainable bone implant in addition to controlled drug delivery.
Keywords: antibiotics; cytotoxicity; fibroblast; in vitro; nanocrystalline; osteomyelitis.
Figures








Similar articles
-
Development of an osteoconductive PCL-PDIPF-hydroxyapatite composite scaffold for bone tissue engineering.J Tissue Eng Regen Med. 2011 Jun;5(6):e126-35. doi: 10.1002/term.394. Epub 2011 Feb 10. J Tissue Eng Regen Med. 2011. PMID: 21312338
-
Hybrid hydroxyapatite nanoparticles-loaded PCL/GE blend fibers for bone tissue engineering.J Biomater Sci Polym Ed. 2013;24(5):520-38. doi: 10.1080/09205063.2012.697696. Epub 2012 Aug 13. J Biomater Sci Polym Ed. 2013. PMID: 23565865
-
O2/Ar Plasma Treatment for Enhancing the Biocompatibility of Hydroxyapatite Nanopowder and Polycaprolactone Composite Film.J Nanosci Nanotechnol. 2015 Aug;15(8):6048-52. doi: 10.1166/jnn.2015.10448. J Nanosci Nanotechnol. 2015. PMID: 26369196
-
Hydroxyapatite/collagen bone-like nanocomposite.Biol Pharm Bull. 2013;36(11):1666-9. doi: 10.1248/bpb.b13-00460. Biol Pharm Bull. 2013. PMID: 24189409 Review.
-
Polymeric implants with drug-releasing capabilities: a mapping review of laboratory research.Drug Dev Ind Pharm. 2021 Oct;47(10):1535-1545. doi: 10.1080/03639045.2022.2043354. Epub 2022 Feb 27. Drug Dev Ind Pharm. 2021. PMID: 35171071 Review.
Cited by
-
Hydroxyapatite-Based Natural Biopolymer Composite for Tissue Regeneration.Materials (Basel). 2024 Aug 20;17(16):4117. doi: 10.3390/ma17164117. Materials (Basel). 2024. PMID: 39203295 Free PMC article. Review.
-
Recent advances in the local antibiotics delivery systems for management of osteomyelitis.Drug Deliv. 2021 Dec;28(1):2392-2414. doi: 10.1080/10717544.2021.1998246. Drug Deliv. 2021. PMID: 34755579 Free PMC article. Review.
-
Levofloxacin loaded mesoporous silica microspheres/nano-hydroxyapatite/polyurethane composite scaffold for the treatment of chronic osteomyelitis with bone defects.Sci Rep. 2017 Feb 2;7:41808. doi: 10.1038/srep41808. Sci Rep. 2017. PMID: 28150731 Free PMC article.
-
An effective antimicrobial complex of nanoscale β-cyclodextrin and ciprofloxacin conjugated to a cell adhesive dipeptide.RSC Adv. 2022 Dec 15;12(54):35383-35395. doi: 10.1039/d2ra05822g. eCollection 2022 Dec 6. RSC Adv. 2022. PMID: 36544467 Free PMC article.
-
Drug Loaded 3D-Printed Poly(ε-Caprolactone) Scaffolds for Local Antibacterial or Anti-Inflammatory Treatment in Bone Regeneration.Polymers (Basel). 2023 Sep 30;15(19):3957. doi: 10.3390/polym15193957. Polymers (Basel). 2023. PMID: 37836006 Free PMC article.
References
-
- Schlickewei CW, Yarar S, Rueger JM. Eluting antibiotic bone graft substitutes for the treatment of osteomyelitis in long bones. A review: evidence for their use? Orthop Res Rev. 2014;6:71–79.
-
- Hutmacher DW. Scaffolds in tissue engineering bone and cartilage. Biomaterials. 2000;21(24):2529–2543. - PubMed
-
- Kim HW, Knowles JC, Kim HE. Hydroxyapatite/poly(epsilon-caprolactone) composite coatings on hydroxyapatite porous bone scaffold for drug delivery. Biomaterials. 2004;25(7–8):1279–1287. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

