Diminished expression of CRHR2 in human colon cancer promotes tumor growth and EMT via persistent IL-6/Stat3 signaling
- PMID: 26495412
- PMCID: PMC4610032
- DOI: 10.1016/j.jcmgh.2015.08.001
Diminished expression of CRHR2 in human colon cancer promotes tumor growth and EMT via persistent IL-6/Stat3 signaling
Abstract
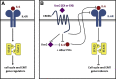
Background & aims: Chronic inflammation promotes development and progression of colorectal cancer (CRC). We explored the distribution of Corticotropin-Releasing-Hormone (CRH)-family of receptors and ligands in CRC and their contribution in tumor growth and oncogenic EMT.
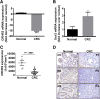
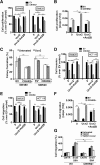
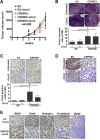
Methods: mRNA expression of CRH-family members was analyzed in CRC (N=56) and control (N=46) samples, 7 CRC cell lines and normal NCM460 cells. Immunohistochemical detection of CRHR2 was performed in 20 CRC and 5 normal tissues. Cell proliferation, migration and invasion were compared between Urocortin-2 (Ucn2)-stimulated parental and CRHR2-overexpressing (CRHR2+) cells in absence or presence of IL-6. CRHR2/Ucn2-targeted effects on tumor growth and EMT were validated in SW620-xenograft mouse models.
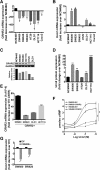
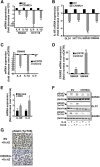
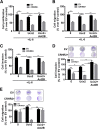
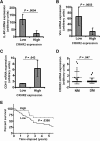
Results: CRC tissues and cell lines showed decreased mRNA and protein CRHR2 expression compared to controls and NCM460, respectively. The opposite trend was shown for Ucn2. CRHR2/Ucn2 signaling inhibited cell proliferation, migration, invasion and colony formation in CRC-CRHR2+ cells. In vivo, SW620-CRHR2+ xenografts showed decreased growth, reduced expression of EMT-inducers and elevated levels of EMT-suppressors. IL-1b, IL-6 and IL-6R mRNAs where diminished in CRC-CRHR2+ cells, while CRHR2/Ucn2 signaling inhibited IL-6-mediated Stat3 activation, invasion, migration and expression of downstream targets acting as cell cycle- and EMT-inducers. Expression of cell cycle- and EMT-suppressors was augmented in IL-6/Ucn2-stimulated CRHR2+ cells. In patients, CRHR2 mRNA expression was inversely correlated with IL-6R and vimentin levels and metastasis occurrence, while positively associated with E-cadherin expression and overall survival.
Conclusions: CRHR2 downregulation in CRC supports tumor expansion and spread through maintaining persistent inflammation and constitutive Stat3 activation. CRHR2low CRC phenotypes are associated with higher risk for distant metastases and poor clinical outcomes.
Keywords: colorectal cancer; inflammation; metastasis; neuropeptides.
Conflict of interest statement
No conflicts of interest exist
Figures

Similar articles
-
CRHR2/Ucn2 signaling is a novel regulator of miR-7/YY1/Fas circuitry contributing to reversal of colorectal cancer cell resistance to Fas-mediated apoptosis.Int J Cancer. 2018 Jan 15;142(2):334-346. doi: 10.1002/ijc.31064. Epub 2017 Oct 12. Int J Cancer. 2018. PMID: 28929494 Free PMC article.
-
Urocortin 2 But Not Urocortin 3 Promotes the Synaptic Formation in Hipppocampal Neurons via Induction of NGF Production by Astrocytes.Endocrinology. 2016 Mar;157(3):1200-10. doi: 10.1210/en.2015-1812. Epub 2015 Dec 29. Endocrinology. 2016. PMID: 26713785
-
Corticotropin-releasing hormone receptors mediate apoptosis via cytosolic calcium-dependent phospholipase A₂ and migration in prostate cancer cell RM-1.J Mol Endocrinol. 2014 Apr 28;52(3):255-67. doi: 10.1530/JME-13-0270. Print 2014 Jun. J Mol Endocrinol. 2014. PMID: 24776847
-
The Neuropeptide System and Colorectal Cancer Liver Metastases: Mechanisms and Management.Int J Mol Sci. 2020 May 15;21(10):3494. doi: 10.3390/ijms21103494. Int J Mol Sci. 2020. PMID: 32429087 Free PMC article. Review.
-
Progress in Understanding the IL-6/STAT3 Pathway in Colorectal Cancer.Onco Targets Ther. 2020 Dec 21;13:13023-13032. doi: 10.2147/OTT.S278013. eCollection 2020. Onco Targets Ther. 2020. PMID: 33376351 Free PMC article. Review.
Cited by
-
Identification of novel enriched recurrent chimeric COL7A1-UCN2 in human laryngeal cancer samples using deep sequencing.BMC Cancer. 2018 Mar 2;18(1):248. doi: 10.1186/s12885-018-4161-8. BMC Cancer. 2018. PMID: 29499655 Free PMC article.
-
MicroRNA-Assisted Hormone Cell Signaling in Colorectal Cancer Resistance.Cells. 2020 Dec 30;10(1):39. doi: 10.3390/cells10010039. Cells. 2020. PMID: 33396628 Free PMC article. Review.
-
PIAS1 inhibited the metastasis of gastric cancer cell by epithelial-mesenchymal transition regulation within the inflammatory microenvironment.Oncol Lett. 2018 Mar;15(3):3828-3837. doi: 10.3892/ol.2018.7811. Epub 2018 Jan 16. Oncol Lett. 2018. PMID: 29556278 Free PMC article.
-
Methylation Status of Corticotropin-Releasing Factor (CRF) Receptor Genes in Colorectal Cancer.J Clin Med. 2021 Jun 18;10(12):2680. doi: 10.3390/jcm10122680. J Clin Med. 2021. PMID: 34207031 Free PMC article.
-
Role of G protein-coupled receptors-microRNA interactions in gastrointestinal pathophysiology.Am J Physiol Gastrointest Liver Physiol. 2017 Nov 1;313(5):G361-G372. doi: 10.1152/ajpgi.00144.2017. Epub 2017 Aug 3. Am J Physiol Gastrointest Liver Physiol. 2017. PMID: 28774868 Free PMC article. Review.
References
-
- Terzic J., Grivennikov S., Karin E. Inflammation and colon cancer. Gastroenterology. 2010;138:2101–2114.e5. - PubMed
-
- Paschos K.A., Kolios G., Chatzaki E. The corticotropin-releasing factor system in inflammatory bowel disease: prospects for new therapeutic approaches. Drug Discov Today. 2009;14:713–720. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
