Inhibition of iNOS activity enhances the anti-tumor effects of alpha-galactosylceramide in established murine cancer model
- PMID: 26496031
- PMCID: PMC4747194
- DOI: 10.18632/oncotarget.6172
Inhibition of iNOS activity enhances the anti-tumor effects of alpha-galactosylceramide in established murine cancer model
Abstract
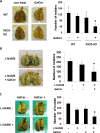
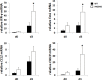
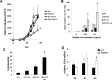
Alpha-garactosylceramide (GalCer) has been shown to have anti-tumor effect in the basic research and clinical studies. However, anti-tumor effect of GalCer is limited. The administration of GalCer increases the production of IFN-γ which is involved in the suppression of tumor growth. On the other hand, the enhancement of IFN-γ production increases immunosuppressive factors such as nitric oxide. This suppressive action might impair the anti-tumor effect of GalCer. In the present study, we evaluated the anti-tumor effect of GalCer in the absence of inducible nitric oxide synthase (iNOS). In lung metastatic model, the number of tumor nodules in the lung of iNOS-KO mice treated with GalCer was significantly reduced compared with that of WT mice treated with GalCer. Moreover, L-NAME, which is the inhibitor for iNOS, enhanced the anti-tumor effect of GalCer in lung metastatic model. The frequency of CD8+ cells in bronchoalveolar lavage fluid increased in iNOS-KO mice treated with GalCer. The administration of GalCer increased the frequency of myeloid-derived suppressor cells (MDSCs) in the lung from tumor-bearing WT mice, but the increase of MDSCs in the lung was not induced in iNOS-KO mice. The subcutaneous tumor experiments revealed that the administration of GalCer in the absence of iNOS expression significantly enhanced the induction of tumor antigen-specific response. Finally, our results indicated that the inhibition of iNOS expression could enhance the therapeutic efficacy of GalCer via the increase of tumor antigen-specific immune response and the suppression of MDSCs.
Keywords: MDSC; alpha-garactosylceramide; cancer immunotherapy; induced nitric oxide synthase; tumor antigen-specific immune response.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



Similar articles
-
α-Galactosylceramide suppresses murine eosinophil production through interferon-γ-dependent induction of NO synthase and CD95.Br J Pharmacol. 2015 Jul;172(13):3313-25. doi: 10.1111/bph.13126. Epub 2015 Apr 24. Br J Pharmacol. 2015. PMID: 25752588 Free PMC article.
-
iNOS expression in CD4+ T cells limits Treg induction by repressing TGFβ1: combined iNOS inhibition and Treg depletion unmask endogenous antitumor immunity.Clin Cancer Res. 2014 Dec 15;20(24):6439-51. doi: 10.1158/1078-0432.CCR-13-3409. Epub 2014 Oct 2. Clin Cancer Res. 2014. PMID: 25278453
-
Combination therapy with α-galactosylceramide and a Toll-like receptor agonist exerts an augmented suppressive effect on lung tumor metastasis in a mouse model.Oncol Rep. 2015 Feb;33(2):826-32. doi: 10.3892/or.2014.3634. Epub 2014 Nov 28. Oncol Rep. 2015. PMID: 25434827
-
Targeting iNOS As a Valuable Strategy for the Therapy of Glioma.ChemMedChem. 2020 Feb 17;15(4):339-344. doi: 10.1002/cmdc.201900580. Epub 2020 Jan 22. ChemMedChem. 2020. PMID: 31851765 Review.
-
[Clinical Application of Immunosuppressive Factors in Cancer Diagnosis and Therapy].Rinsho Byori. 2016 Dec;64(12):1360-1366. Rinsho Byori. 2016. PMID: 30653899 Review. Japanese.
Cited by
-
Biological Roles of Aberrantly Expressed Glycosphingolipids and Related Enzymes in Human Cancer Development and Progression.Front Physiol. 2018 May 3;9:466. doi: 10.3389/fphys.2018.00466. eCollection 2018. Front Physiol. 2018. PMID: 29773994 Free PMC article. Review.
-
SETD1B Activates iNOS Expression in Myeloid-Derived Suppressor Cells.Cancer Res. 2017 Jun 1;77(11):2834-2843. doi: 10.1158/0008-5472.CAN-16-2238. Epub 2017 Apr 5. Cancer Res. 2017. PMID: 28381543 Free PMC article.
-
α-GalCer and iNKT Cell-Based Cancer Immunotherapy: Realizing the Therapeutic Potentials.Front Immunol. 2019 Jun 6;10:1126. doi: 10.3389/fimmu.2019.01126. eCollection 2019. Front Immunol. 2019. PMID: 31244823 Free PMC article. Review.
-
Regulation of T cells by myeloid-derived suppressor cells: emerging immunosuppressor in lung cancer.Discov Oncol. 2023 Oct 19;14(1):185. doi: 10.1007/s12672-023-00793-1. Discov Oncol. 2023. PMID: 37857728 Free PMC article. Review.
-
Calycosin Induces Gastric Cancer Cell Apoptosis via the ROS-Mediated MAPK/STAT3/NF-κB Pathway.Onco Targets Ther. 2021 Apr 12;14:2505-2517. doi: 10.2147/OTT.S292388. eCollection 2021. Onco Targets Ther. 2021. PMID: 33883905 Free PMC article.
References
-
- Ito H, Ando K, Nakayama T, Taniguchi M, Ezaki T, Saito K, Takemura M, Sekikawa K, Imawari M, Seishima M, Moriwaki H. Role of Valpha 14 NKT cells in the development of impaired liver regeneration in vivo. Hepatology. 2003;38:1116–1124. - PubMed
-
- Ito H, Koide N, Hassan F, Islam S, Tumurkhuu G, Mori I, Yoshida T, Kakumu S, Moriwaki H, Yokochi T. Lethal endotoxic shock using alpha-galactosylceramide sensitization as a new experimental model of septic shock. Lab Invest. 2006;86:254–261. - PubMed
-
- Ito H, Ando K, Ishikawa T, Nakayama T, Taniguchi M, Saito K, Imawari M, Moriwaki H, Yokochi T, Kakumu S, Seishima M. Role of Valpha14+ NKT cells in the development of Hepatitis B virus-specific CTL: activation of Valpha14+ NKT cells promotes the breakage of CTL tolerance. International immunology. 2008;20:869–879. - PubMed
-
- Fuji N, Ueda Y, Fujiwara H, Toh T, Yoshimura T, Yamagishi H. Antitumor effect of alpha-galactosylceramide (KRN7000) on spontaneous hepatic metastases requires endogenous interleukin 12 in the liver. Clinical cancer research : an official journal of the American Association for Cancer Research. 2000;6:3380–3387. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

