The protective role of montelukast against intestinal ischemia-reperfusion injury in rats
- PMID: 26497763
- PMCID: PMC4620564
- DOI: 10.1038/srep15787
The protective role of montelukast against intestinal ischemia-reperfusion injury in rats
Abstract
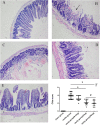
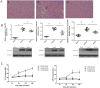
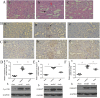
Several drugs are effective in attenuating intestinal ischemia-reperfusion injury (IRI); however little is known about the effect of montelukast. Fifty rats were randomly assigned to 3 groups: model group (operation with clamping), sham group (operation without clamping), and study group (operation with clamping and 0.2, 2 and 20 mg/kg montelukast pretreatment). Intestinal ischemia-reperfusion was performed by occlusion (clamping) of the arteria mesenterica anterior for 45 min, followed by 24 h reperfusion. Intestinal IRI in the model group led to severe damage of the intestinal mucosa, liver and kidney. The Chiu scores of the intestines from the study group (2 and 20 mg/kg) were lower than that of the model group. Intestinal IRI induced a marked increase in CysLTR1, Caspase-8 and -9 expression in intestine, liver and kidney, which were markedly reduced by preconditioning with 2 mg/kg montelukast. Preconditioning with 2 g/kg montelukast significantly attenuated hepatic tissue injury and kidney damage, and decreased plasma interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) levels in plasma after intestinal IRI. In conclusion, preconditioning with montelukast could attenuate intestinal IRI and the subsequent systemic inflammatory response in rats.
Figures




Similar articles
-
Resolvin D1 protects against hepatic ischemia/reperfusion injury in rats.Int Immunopharmacol. 2015 Sep;28(1):322-7. doi: 10.1016/j.intimp.2015.06.017. Epub 2015 Jun 25. Int Immunopharmacol. 2015. PMID: 26118631
-
Protective Effects of Geniposide on Hepatic Ischemia/Reperfusion Injury.Transplant Proc. 2017 Jul-Aug;49(6):1455-1460. doi: 10.1016/j.transproceed.2017.02.063. Transplant Proc. 2017. PMID: 28736023
-
Protective potential of montelukast against hepatic ischemia/reperfusion injury in rats.J Surg Res. 2010 Mar;159(1):588-94. doi: 10.1016/j.jss.2008.08.006. Epub 2008 Sep 7. J Surg Res. 2010. PMID: 19515388
-
Melatonin and its protective role in attenuating warm or cold hepatic ischaemia/reperfusion injury.Cell Prolif. 2021 Apr;54(4):e13021. doi: 10.1111/cpr.13021. Epub 2021 Mar 10. Cell Prolif. 2021. PMID: 33751704 Free PMC article. Review.
-
Global consequences of liver ischemia/reperfusion injury.Oxid Med Cell Longev. 2014;2014:906965. doi: 10.1155/2014/906965. Epub 2014 Apr 1. Oxid Med Cell Longev. 2014. PMID: 24799983 Free PMC article. Review.
Cited by
-
Preventative effect of montelukast in mild to moderate contrast-induced acute kidney injury in rats via NADPH oxidase 4, p22phox and nuclear factor kappa-B expressions.Int Urol Nephrol. 2025 Jul;57(7):2313-2325. doi: 10.1007/s11255-025-04378-5. Epub 2025 Feb 21. Int Urol Nephrol. 2025. PMID: 39982657
-
Montelukast Prevents Mice Against Acetaminophen-Induced Liver Injury.Front Pharmacol. 2019 Sep 18;10:1070. doi: 10.3389/fphar.2019.01070. eCollection 2019. Front Pharmacol. 2019. PMID: 31620001 Free PMC article.
-
The anti-inflammatory effect of montelukast, a cysteinyl leukotriene receptor-1 antagonist, against estradiol-induced nonbacterial inflammation in the rat prostate.Naunyn Schmiedebergs Arch Pharmacol. 2017 Feb;390(2):197-205. doi: 10.1007/s00210-016-1325-4. Epub 2016 Dec 1. Naunyn Schmiedebergs Arch Pharmacol. 2017. PMID: 27909742
-
Pretreatment with the ALDH2 agonist Alda-1 reduces intestinal injury induced by ischaemia and reperfusion in mice.Clin Sci (Lond). 2017 Jun 1;131(11):1123-1136. doi: 10.1042/CS20170074. Epub 2017 Mar 21. Clin Sci (Lond). 2017. PMID: 28325855 Free PMC article.
-
Leukotrienes and kidney diseases.Curr Opin Nephrol Hypertens. 2018 Jan;27(1):42-48. doi: 10.1097/MNH.0000000000000381. Curr Opin Nephrol Hypertens. 2018. PMID: 29059080 Free PMC article. Review.
References
-
- Huang X. J. et al. Activation of CysLT receptors induces astrocyte proliferation and death after oxygen-glucose deprivation. Glia 56, 27–37 (2008). - PubMed
-
- Pergel A. et al. Anti-inflammatory and antioxidant effects of infliximab in a rat model of intestinal ischemia/reperfusion injury. Toxicol Ind Health 28, 923–932 (2012). - PubMed
-
- Hagar H. H. & Abd El Tawab R. Cysteinyl leukotriene receptor antagonism alleviates renal injury induced by ischemia-reperfusion in rats. J Surg Res 178, e25–34 (2012). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

