Dramatic Response of BRAF V600E Mutant Papillary Craniopharyngioma to Targeted Therapy
- PMID: 26498373
- PMCID: PMC4862417
- DOI: 10.1093/jnci/djv310
Dramatic Response of BRAF V600E Mutant Papillary Craniopharyngioma to Targeted Therapy
Abstract
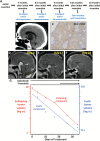
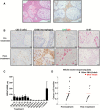
We recently reported that BRAF V600E is the principal oncogenic driver of papillary craniopharyngioma, a highly morbid intracranial tumor commonly refractory to treatment. Here, we describe our treatment of a man age 39 years with multiply recurrent BRAF V600E craniopharyngioma using dabrafenib (150mg, orally twice daily) and trametinib (2mg, orally twice daily). After 35 days of treatment, tumor volume was reduced by 85%. Mutations that commonly mediate resistance to MAPK pathway inhibition were not detected in a post-treatment sample by whole exome sequencing. A blood-based BRAF V600E assay detected circulating BRAF V600E in the patient's blood. Re-evaluation of the existing management paradigms for craniopharyngioma is warranted, as patient morbidity might be reduced by noninvasive mutation testing and neoadjuvant-targeted treatment.
© The Author 2015. Published by Oxford University Press. All rights reserved. For Permissions, please e-mail: journals.permissions@oup.com.
Figures
References
-
- Crotty TB, Scheithauer BW, Young WF, Jr, et al. Papillary craniopharyngioma: a clinicopathological study of 48 cases. J Neurosurg. 1995;83 (2):206–214. - PubMed
-
- Ascierto PA, Minor D, Ribas A, et al. Phase II Trial (BREAK-2) of the BRAF Inhibitor Dabrafenib (GSK2118436) in Patients With Metastatic Melanoma. J Clin Oncol. 2013;31(26):3205–3211. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

